BISC209: Roles of soil Microbes
Examining Roles of Soil Bacteria in an Ecosystem
The following tests allow you to examine some of the catabolic abilities of your bacterial isolates. It is unlikely that you will be able to test every isolate you have nursed along during the preceeding weeks with every one of these tests. Please select tests that can be used, if positive, as evidence for some suspected role you think the bacteria from a particular enrichment group might possess or that could be an unexpected, yet important, role for one of your isolate. Select isolates from the greatest variety of enrichment media.
Glucose catabolism - Fermentation Media
Screening for carbohydrate catabolism
Glucose is a simple sugar that can enter cells directly; therefore, it is a common energy source and preferred by many organisms. (Remember your BISC110 study of glucose repression: a common way to downregulate production of enyzmes that allow catabolism of other sugars like lactose.) Some bacteria can only catabolize glucose in the presence of oxygen (oxidative catabolism), while others can also (or only) ferment the glucose, breaking it down to acidic products in the presence and/or absence of oxygen. Based on the outcome of the OF-glucose test bacteria can be separated into distinct groups: strict aerobes (the oxidation of glucose) and facultative aerobes (an anaerobic process).
OF Glucose Medium is useful in screening for the ability of organisms to oxidize and/or ferment sugars. The medium is composed of a high concentration of glucose (carbohydrate) and a low concentration of peptone. The peptone supports the growth of bacteria unable to oxidize or ferment the carbohydrate. The medium is used as a semi-solid agar deep. ([Original paper] http://www.ncbi.nlm.nih.gov/pmc/articles/PMC357086/: Hugh, Rudolph and Einar Leifson. 1953. Journal of Bacteriology. 66(1): 24-26.)
Glucose is a common chemoheterotrophic bacterial energy source either by fermentation and/or respiration. Glucose used in respiration is observed only if there is no fermentation because of the large amount of acids produced by glucose fermentation masking the respiratory products. The respiratory pathway is seen only in strict aerobes that use glucose to respire and cannot ferment. This link is helpful to interpret OF-Glucose: http://www.jlindquist.net/generalmicro/dfnewglucoseofpage.html].
Glucose can also be used as a source of carbon, however because there are a large number of other potential carbon sources (amino acids as well as glucose) in most media, whether or not glucose is used as a carbon source cannot be directly determined from the reactions seen in these media.
Peptone is a commonly-used medium ingredient which mainly supplies amino acids (sources of nitrogen, carbon, sulfur and energy for many bacteria). A minimum amount of peptone is included in OF-Glucose medium to prevent ammonium (alkaline) released from deamination of amino acids from neutralizing the acid production we are trying to observe.
pH indicator: Brom-thymol blue turns yellow with a net acidic pH and blue with a net alkaline pH.
OF Glucose medium recipe: peptone, 0.2 per cent; NaCl, 0.5 per cent; K2HPO4, 0.03 per cent; agar, 0.3 per cent; bromthymol blue, 0.003 per cent; glucose, 1.0 per cent; pH 7.1.
OF-Glucose Test Procedure:
Observe and record the color of the uninoculated tubes of OF Glucose semi-solid deeps.
Label one screw cap tube and one loosely capped tube of this medium.
Using an inoculating needle (NOT your loop!), inoculate part of an isolated colony from a pure culture to each tube by placing the needle with the bacteria straight down into the middle of the tube and pulling it out in the same line. DO NOT try to mix the bacteria into the medium.
Use a Pasteur pipet to layer a few drops of mineral oil on the surface of the medium in the screw capped tube so that the oil blocks the air interface but doesn't mix with the medium.(There is a bottle of mineral oil on the instructor bench.) DO NOT add mineral oil to the surface of the loosely capped tube!
Incubate both tubes of inoculated medium at RT until you can detect significant growth (a few days to a week).
Observe for:
Presence of growth (cloudy/not cloudy at the inoculation line only or spreading away from the inoculation line)
Color change of medium: a change to yellow indicates acidic by-products of glucose catabolism; a change to dark blue indicates alkaline by-products due to the use of peptone, not glucose.
Type of metabolism: 1. oxidative-- where glucose is metabolized only when oxygen is available or 2. fermentative-- where glucose is metabolized in the absence of oxygen.
Observe both tubes and compare to an uninoculated tube. Note whether or not the growth pattern of bacteria in the screw-capped tube has moved away from the inoculation line, indicating a motile organism. More importantly, a change in the color of the bromothymol blue indicator to yellow indicates the ability of that organism to breakdown glucose to acidic by-products. If both the loosely capped and screw cap tube are yellow, the organism is considered a facultative anaerobe because it is able to ferment the carbohydrate. The presence of an oxidative pathway only indicates that the organism is a strict aerobe. Oxidative only breakdown of glucose is indicated by a change to yellow only in the upper portion of the loosely capped tube and no yellow color in the tightly capped tube. If only the top portion of the medium in the loosely capped tube turns from greenish to a blue color, it means the organism is also a strict aerobe, although it cannot oxidize or ferment glucose. Note that the color change to blue comes from the ability to break down the peptone to produce an alkaline product aerobically.
If an isolate is able to ferment glucose in OF-glucose medium, try testing its ability to ferment different sugars using the protocol for Carbohydrate Fermentation Medium. Your lab instructor will advise you as to what carbohydrates are available for testing.
Cellulose degradation
Carbon is a basic component of organic molecules. Its availability for animals and plants is critical. Many soil bacteria are able to accomplish CO2 recycling of an abudant source, cellulose. Plants store large amounts of carbon as cellulose but cellulose is not easily broken down by plants or animals. Bacteria that have the enzyme cellulase play an crucial recycling role in breaking down such plant material into compost. They also make valuable by-products available for other microorganisms while, eventually, cycling the carbon for plants and animals to use again.
Materials:
Soil bacteria to test, grown in an appropriate broth culture, preferable in log phase;
Melted nutrient agar,
sterile petri dishes,
leaf or plant material discs, taken from the plants near the location of soil sampling in the Greenhouse,
Sterile swabs
Protocol: One plate for 2 bacteria to be tested
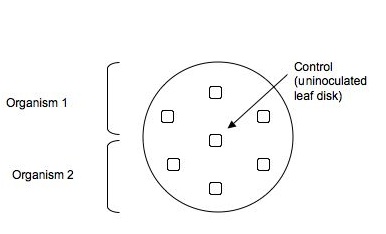
- Go down to the Greenhouse to your habitat under investigation. Put on gloves and collect 3 different plant's leaves (2-3 leaves per plant) from those around your soil sampling site. Choose mature, living leaves from inconspicuous places at the bottom of the plant. Put the leaves in a sandwich bag and return to the lab after you have recorded the name of the plants and their location. When back in the lab, wearing gloves and holding an autoclaved microfuge tube, open the lid of the microfuge tube and position it so that the tube is below the leaf and the top is above it. Punch out 7 leaf discs for each plate your teams plan to set up. (8 plates/habitat if there are 16 isolates being characterize -4 per team member.) Punch the disks of different leaf types into separate microfuge tubes.
- Pour about 15ml of melted nutrient agar into labeled empty sterile Petri plates. Two isolates can be tested on each plate. (See illustration below.) If your group is testing 16 isolates, someone in the group should make 8 plates (as described and pictured below) for everyone to use.
-
Use the sharp end of micropipet tip to spear, separate, and push leaf discs into the agar, pressing gently so that the leaf pieces are on the surface of the agar, but deep enough to be held in place once the agar hardens. Organize the leaf disks so that three disks are positioned in the top third of the plate, one is alone in the center and three disks are in the bottom third of each plate.
- Allow the agar to finish solidifying (about 10 more minutes).
- If you have a 24 or 48 hour broth culture of your isolate, use a sterile swab to inoculate your organism onto the center of the three leaf disks on half the plate, inoculate a second organism on the other three disks, the center disk is for comparison. If your organism is on solid media, rather than broth, take a small amount of pure culture and suspend it in sterile water until the water is visibly but only slightly cloudy. Use that to swab an inoculum on the center of each leaf disk. Be gentle so you don't displace the leaf disks. Discard the swabs in your autoclave bag.
- Incubate plates at RT after you seal the edges with parafilm and put them all in a zip lock bag. Check at least once a week for evidence of leaf breakdown or for dehydration. You may add a few drops of sterile water if the plates appear to be drying out. Keep checking over at least 4 weeks.
- When you think you see evidence of leaf breakdown use a dissecting scope to see more details and draw and record your observations. You might try scoring the degree of breakdown using a 1-5 scale (1=no change in leaf disk to 5= leaf tissue highly digested).
-
If an organism that you expect will degrade cellulose is growing poorly on nutrient agar, oatmeal agar (a cellulitic media) might be a better choice, but unfortunately it is not available for S10.(see: Enrichment Media for Soil Bacterial from Mixed Populations )
Ammonification by soil microbes
As plants and animals die and decay in the soil, the nitrogen stored in organic molecules, (such as the amino acids of proteins) needs to be retrieved. Some soil bacteria, like the Ammonia Nitrifyers we isolated, are able to convert proteins to amino acids and then digest the amino acids yielding ammonia in a process known as ammonification. It is an important role of some soil bacteria because the ammonia produced can be used by that organism or other soil bacteria to convert ammonia to either nitrite, or nitrate, or both. Still other bacteria can convert ammonia to Nitrogen gas as we saw when selecting and enriching for the Hyphomicrobia (denitrifying methylotrophs). An others, like the Azotobacteriaceae, fix atmospheric Nitrogen forming 2 moles of ammonia from one mole of nitrogen, N2 + 8H+ + 8e- + 16 ATP = 2NH3 + H2 + 16ADP + 16 Pi. Select only isolates you believe are Azotobacteria or Ammonia nitrification to perform the following tests to assess your their ability to convert protein to ammonia or nitrate. If, from your reading, you believe organisms isolated from any of the other protocols might be breaking down proteins to ammonia or nitrate, please ask your instructor if you can run the test. We don't want to waste the reagents on organisms unlikely to be positive. Also, it is possible that we will obtain falsely low results because of the buffering capacity of the medium.
(Adapted from: Alacamo, Edward. 2001. Laboratory Fundamentals of Microbiology 6th ed. Jones and Bartlett Publishers. Sudbury, MA. p. 268-269.)
Materials:
Inoculate the organism(s) to be tested in sterile peptone broth media ( 2% BactoPeptone, 0.5% sodium chloride and 1.5% agar. Adjust final pH to 7.2-7.4). Inoculate and incubate plates at 35 ± 2°C for 18-48 hours. in tubes in log phase or fresh, depression plates, Nessler's reagent substitutes (2010- use aquarium water test reagents for ammonia, nitrite and nitrate), glass slides (non-sterile)
Testing PROTOCOL for Ammonia (NH3/NH4) and/or Nitrate NO3- production:
- Label a "control" tube of sterile peptone broth media. Don't do anything to it. You will use it for comparison later.
For each bacterial isolate to be tested:
- Inoculate a separate tube of sterile peptone broth with a loop full of pure culture of each of the bacterial isolates to be tested.
- Incubate all the tubes (including the uninoculated control) at 30C for 48 hours
TEST for Nitrate and Ammonia:
(NOTE: Because the traditional test uses a highly toxic reagent, Nessler's reagent, which has a substantial mercury component, we have available instead typical aquarium water testing reagents that you can buy at pet stores. We have different reagents for testing for nitrate and ammonia. The Ammonia test kit reads the total ammonia level in parts per million (ppm), equivalent to mg/L if in the range 0-8. The nitrate kit reads total Nitrate in parts per million, equivalent to mg/L in the range 0-160.
Obtain (2) 13 x100mm glass test tubes (not sterile) and several small pieces of parafilm from the equipment area and use your P1000 to pipet 0.5 ml of your broth culture into each tube. Pipet 2 ml of deionized water (does not have to be sterile) into the same tubes. Label one tube Ammonia and the other Nitrate.
Mix the reagents by inverting the bottles.
AMMONIA TEST
NITRATE TEST
After the color development period has ended, compare the color of both the ammonia test and the nitrate test to the uninoculated broth culture and to the FRESHWATER color charts (not the marine ones) found in the reagent box. Make sure you hold the tubes over the white section of the card located next to the colored boxes in a well lighted area. Use the color charts to evaluate color changes indicative of nitrate or ammonia. Make sure that you return the color charts and the reagents to the reagent box and return the boxes to the equipment area so that other students can find the test reagents and evaluation tools!
If you obtain a negative result, use the remaining inoculated broth cuture to subculture a new broth and incubate the new peptone broth culture for a week rather than for 48 hours and retest.
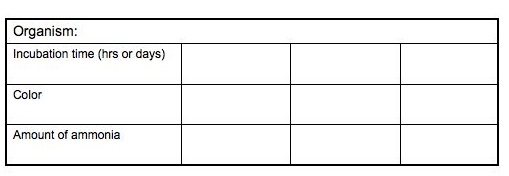
Amount of Ammonia Present/ Incubation Time
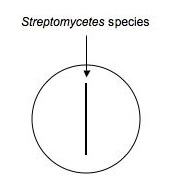
Antibiotic production
Many microbes secrete antimicrobial compounds to help them compete with other microorganisms for habitat. Some of the bacteria that are common antibiotic producers are the Actinomycetes (including Streptomycetes species), many of the Bacillus species, and the fruiting myxobacteria, to name just a few among many, many antibiotic producing bacteria. You can also test for the opposite: the sensitivity of your soil organisms (or known stock bacteria) to manufactured or secreted antibiotics.
(This testing will take 3 weeks.)
Week 1:
Identify how many potential antibiotic producers you might have. Definitely test any isolates that are likely to be Actinomycetes, Myxobacteria, or Bacillus. It might be wise to test all 4 of your isolates since the soil is the main source of microbes that supply the world's antibiotics. It's possible that you might discover the next great antimicrobial drug and get very rich by selling the patent for your discovery to a drug company. Remember that the discovery of penicillin was completely accidental.
Using aspectic technique, transfer a small part of an isolated colony (possible Streptomyces, etc.) that's likely to be an antibiotic producer to your loop. Drag the loop (as shown below) down the middle of a plate of nutrient agar. Make a second plate exactly like the first for each isolate to be tested. Label them carefully and incubate the plates for ~1 week at RT.
Week 2
TO BE PROVIDED in LAB 6:
3 fresh cultures of Eschericia coli (Gram negative), Staphylococcus epidermidis (Gram positive) and Micrococcus luteus (Gram positive) grown in nutrient broth.
NEED TO MAKE (Optional): If you want to test any of your isolates for sensitivity to an antibiotic producer, start a log phase broth culture in appropriate media so that it will be ready to use in LAB 6.
PROTOCOL
- Use a sterile swab and aseptically apply a line of inoculation of each of the provided broth cultures of : E. coli, Micrococcus, and S. epidermidis. as shown below. Draw a line perpendicular to the antibiotic producer's (Streptomyces) inoculation. Be careful not to touch the antibiotic producer's growth. Using the same swabs, inoculate a new NA plate (one plate for all three cultures)by making a line across the plate for E. coli, Micrococcus and S. epidermidis. Incubate this plate along with your test plate. It will serve as a control to make sure that lack of growth is due to antibiotic sensitivity and not to no living cells in the inoculum.
-

- Repeat this procedure on another plate using broth cultures made of any of your isolates that you want to test for sensitivity to your antibiotic producers,
- Label a template in your notebook with which organism is where on each plate.
- Incubate for another week.
Week 3
- Examine the plate and look for evidence of inhibition of growth of "test" organisms near the antibiotic producer's midline streak.
- Draw the results and evaluate whether or not there was evidence that an antibiotic was produced by the organism and, if so, which of the bacteria tested were sensitive to it and to what degree. If you found no inhibition of growth, does that mean that your potential antibiotic producer does not secrete an any antimicrobial compounds? Why or why not?
Starch digestion
Starch, a polysaccharide made from glucose, is a plant storage molecule. It is found in two forms: a straight chain form, amylose, and a branched form, amylopectin. Organisms able to break down starch hydrolyze the polysaccharide to maltose (two glucose units) using the enzyme amylase. The disaccharide Maltose is then available for use by many other organisms. The amylase is an exoenzyme secreted by a variety of bacteria (and fungi). The digestion of starch can be observed by flooding an inoculated and incubated plate with Gram's iodine. Amylose will give a deep-blue color, amylopectins give a red to brown color. In many plant storage tissues (e.g potato root) a mix of both amylose and amylopectin are present. If the starch is digested, no color will develop in the areas of starch hydrolysis.
- Use one starch plate divided into 4 labeled and coded quadrants to test the ability of 4 organisms at a time to digest starch.
- Use the inoculating needle or loop to place a tiny amount of growth in one spot in the center of a a quadrant. Do not spread the inoculum at all.
- Incubate the plates until you see a visible amount of growth where each isolate was inoculated.
- Apply Gram's iodine to the plate (from your Gram stain kit) until the surface of the plate is just covered. Pour off any excess iodine in the sink. Look for clear or lighter color zones (halos) in the media around any of your colonies. That light or clear halo is a positive test. Since the medium contains starch, you should see the whole plate turn dark except where the starch digesting enzymes were secreted and broke down the starch around any of the positive organisms. A negative test is indicated by no difference in color around a bacterial colony compared to the color of the medium that is not near any bacterial growth.
Links to Labs
Lab 1
Lab 2
Lab 3
Lab 4
Lab 5
Lab 6
Lab 7
Lab 8
Lab 9
Lab 10
Lab11
Lab 12