SBB10Ntbk-JingLuo: Difference between revisions
No edit summary |
No edit summary |
||
| Line 31: | Line 31: | ||
* Generate the insert | * Generate the insert | ||
* Restriction enzyme digest the insert and vector | * Restriction enzyme digest the insert and vector | ||
* Purify necessary fragments using prepative gel | |||
* Ligate insert and vector together | * Ligate insert and vector together | ||
* Transform | * Transform | ||
Revision as of 23:15, 3 May 2010
Construction of sbb20: FokI Cleavage Domain - <FokI-! PCR gh1000F/gh1001R on BBa_K243001 (261 bp, gp = A) PCR gh1001F/gh1003R on BBa_K243001 (264 bp, gp = B) PCR gh1003F/gh1000R on BBa_K243001 (144 bp, gp = C) --------------------------------------------------- PCR gh1000F/gh1000R on A+B+C (622 bp, EcoRI/BamHI) Digest pBjk2741 (EcoRI/BamHI, 2170+910 L) Product is pBjk2741-sbb20 --------------------------------------------------- gh1000F Forward for part <FokI-! ccagtGAATTCatgAGATCTCAGCTGGTTaaatctgaactggaggag gh1001F Making internal mutation 1+2 cgttggttccccgatcgattatggcgttatcgtggAcacaaaagc gh1001R Making internal mutation 1+2 cgccataatcgatcggggaaccaacggtataaatggcaccGTctggtttac gh1003F Making internal mutation 3 ccatatcaccaatTgcaatggggcagtgctgag gh1003R Making internal mutation 3 ccattgcAattggtgatatgg gh1000R Reverse for part <FokI-! gcaaaGGATCCTTAaaaattgatctcgccattgttg Part: GATCTCAGCTGGTTaaatctgaactggaggagaaaaaatccgagctgcgccacaaactgaaatatgtgcctcacgagtatatcgaactgatcgagatcgcccgtaatagtacccaagaccgtatcctggaaatgaaagtgatggagttcttcatgaaagtctatggctatcgtggcaaacatctgggtggtagccGTAAACCAGACGGTGCCATTTATACcgttggttccccgatcgattatggcgttatcgtggAcacaaaagcgtattctggcggttataatctgccgattggtcaggctgatgagatggaacgttatgtggaagagaatcagacccgtaacaaacatctgaacccgaacgaatggtggaaagtgtatccgtcaagtgtcaccgagttcaaatttctgttcgtgagcggccactttaaaggcaactataaagcccagctgactcgtctgaaccatatcaccaatTgcaatggggcagtgctgagtgttgaggaactgctgatcggtggagaaatgatcaaagcaggcaccctgactctggaagaagttcgccgtaaattCAACAATGGCGAGATCAATTTTTAAG
Construction of sbb35: P9 rep Protein - {rbs.ColE2 RepA}
Part already exists, only requires a EcoR1/BamH1 transfer into pBca9145-Bca1144.
Gel ladder that was used for all the experiments
General Experiment Outline:
- Generate the insert
- Restriction enzyme digest the insert and vector
- Purify necessary fragments using prepative gel
- Ligate insert and vector together
- Transform
- Plate
- Pick colonies and culture
- Miniprep colonies
- Analytical digest mapping
- Sequencing of colony
Inventory (Inside Box C)
- y A(star) - part A for SOEing
- y B(star) - part B for SOEing
- y C(star) - part C for SOEing
- JHL Gel frag - preparative gel fragments of PCR A, B, C dissolved in ADB buffer
- PCR Zc ABC - Zymo gel clean preparative gel fragments of PCR A, B, C
- sbb20 Sew - SOEing with external primers, first attempt
- sbb20 SOE H2O - 2nd round of SOEing of PCR products A, B, C
- sb20 digest zc - Digested w/ EcoR1/BamH1 and zymo cleaned of the Round 2 SOEing reaction
- 20 digest prep gel - sbb20 from preparative gel after digestion
- JHL SOE Pro Zc -
- Xfer digest zc - Eco/Bam transfer digest and zymo cleaned
- MP20 #1 - Miniprepped sbb20 colony from transformed cells w/ part
- MP20 #2 - Miniprepped sbb20 colony from transformed cells w/ part
- MP20 #3 - Miniprepped sbb20 colony from transformed cells w/ part
- MP20 #4 - Miniprepped sbb20 colony from transformed cells w/ part
- sb35 1 - Miniprepped sbb35 colony from transformed cells w/ part
- sb35 2 - Miniprepped sbb35 colony from transformed cells w/ part
- sb35 3 - Miniprepped sbb35 colony from transformed cells w/ part
- sb35 4 - Miniprepped sbb35 colony from transformed cells w/ part
2/17
Part sbb20:
Purpose: Create sbb20 insert -- Using the designed oligos to clone the fragments of sbb20, which will later be SOE'd together.
Protocol:
- Using the 2K55 temperature program, cloned the necessary parts for SOEing using the protocol: Cloning by PCR
Results
- The next step is to run a preparative gel to determine the results of the PCR.
Products labeled:
- y A(star) (sbb20A), y B(star) (sbb20B), y C(star) (sbb20C)
2/22
Part sbb20:
Purpose: Create sbb20 insert -- Identify the presence or not of the appropriate sequence, extracting it and purifying it from the gel.
Protocol:
- Ran a preparative gel on the PCR products from 2/17 (sbb20A,sbb20B,sbb20C)
Results
- Results of the preparative gel looked good. Lanes 9, 10, 11 (Part A, B, C respectively): Gel pic
- ladder
- sbb04A
- sbb04B
- sbb04C
- RH 33A
- RH33B
- blank
- blank
- CD1 <-- Part A
- CD2 <-- Part B
- CD3 <-- Part C
- sbb19A
- sbb19B
- ladder
- Ran a gel purification of the preparative gel fragments using protocol: Zymo gel purification
Products labeled:
- Preparative gel: JHL Gel frag
- Post zymo clean: PCR Zc ABC
2/24
Part sbb20:
Purpose: Create sbb20 insert -- Using the external primers to combine the three parts together into sbb20.
Protocol:
- Ran a SOEing reaction using protocol: SOEing by PCR
Results
- Next step is to run an analytical gel to determine the results of the SOEing reaction.
Products labeled:
- sbb20 sew
3/1
Part sbb20:
Purpose: Create sbb20 insert -- Identify whether or not the SOEing reaction produced the full length sequence.
Protocol:
- Ran an analytical gel
Results
- Results of analytical gel showed SOEing PCR failed to produce any product: Gel pic
- Ladder
- RH33-2K45
- RH33-2K45 w/ DMSO
- sbb02
- sbb01
- sbb03
- JL20 <-- SOEing reaction
- PiggyBacA
- Ladder
- Reattempting the SOEing, by repeating with a modified SOEing PCR to use DMSO or H2O and temperature program 45: SOEing by PCR
Products labeled:
- sbb20 SOE H2O
- sbb20 SOE DMSO
3/3
Part sbb20:
Purpose: Create sbb20 insert -- Identify the presence or not of the appropriate sequence from the SOEing reactions and extract it.
Protocol:
- Ran on preparative gel.
Results
- The full length sequence was present: Gel pic
- Ladder
- sbb20 Digest <-- sbb20 SOE H2O
- sbb20 Digest <-- sbb20 SOE DMSO
- KRM27 1
- KRM27 2
- KRM27 3
- KRM27 4
- KRM16 1
- KRM16 2
- KRM16 3
- KRM16 4
- FK114
- Ladder
- It was extracted and run through gel purification using protocol: Zymo gel purification.
- Digested the product with restriction enzymes using the protocol: EcoR1/BamH1 Digest of PCR products
Products labeled:
- 20 digest prep gel
3/8
Part sbb20:
Purpose: Create the full correct plasmid (sbb20 part + vector) -- Combine the digested insert (sbb20 part) with the vector, transform into cells, and plate.
Protocol:
- Ligated the part with the pBjk2741 vector using protocol: Ligation of EcoR1/BamH1 Digests
- Transformed into JTK086 spec cells using protocol: Transformation by Heat Shock
- GSI's plated out onto spec antibiotic plate.
Results
- The next step is to see if any colonies grow up on the selective resistance plates, if so pick colonies and culture them.
Products labeled:
- JHL sbb20 plate
Part sbb35:
Purpose: Create the full correct plasmid (sbb35 part + vector) -- Transfer the part from one vector to another, starting with digestion of the sbb35 part.
Protocol:
- Began a Eco/Bam transfer using protocol: EcoR1/BamH1 Part Transfer
- stopped after the restriction enzyme digestion that was done using protocol: EcoR1/BamH1 Digest of PCR products
- performed a regular zymo clean up on the part using protocol: Regular zymo cleanup
Results
- The next step is to digest the vector and ligate the two together, whether or not this works will be determined once the cells are miniprepped and a analytical digest is run.
Products labeled:
- Xfer digest zc
3/9
Part sbb20:
Results:
- Colonies did grow and the GSI's picked out colonies and cultured them using protocol: Picking of Colonies
3/10
Part sbb20:
Purpose: Create the full correct plasmid (sbb20 part + vector) -- Extract the DNA from the colonies so that it can be analyzed.
Protocol:
- Minipepped cells using protocol: Macherey-Nagel Minipepped
Results
- Need to determine whether or not the cells successfully took the full plasmid, this will be done next time by analytical gel.
Products labeled:
- MP20 #1, MP20 #2, MP20 #3, and MP20 #4
Part sbb35:
Purpose: Create the full correct plasmid (sbb35 part + vector) -- Combine the insert with the vector, transform into cells, and plate.
Protocol:
- The pBca1256 vector was pre-digested by the GSI's using protocol: EcoR1/BamH1 Digest of PCR products
- Ligated the part with pBca1256 vector instead of pBca9523 using protocol: Ligation of EcoR1/BamH1 Digests
- Transformed into DH108 spec cells using protocol: Transformation by Heat Shock
- Plated out onto spec antibiotic plate
Results
- The next step is to see if any colonies grow up on the selective resistance plates, if so pick colonies and culture them.
Products labeled:
- JHL sbb35
3/11-15
Part sbb35:
- GSI's picked a colony and cultured it according to protocol: Picking of Colonies
3/15
Part sbb20:
Purpose: Create the full correct plasmid (sbb20 part + vector) -- Determine if the transformed cells DNA contain the full plasmid.
Protocol:
- Ran an analytical digest map with a portion of the miniprepped DNA using protocol: Analytical digest mapping
Results
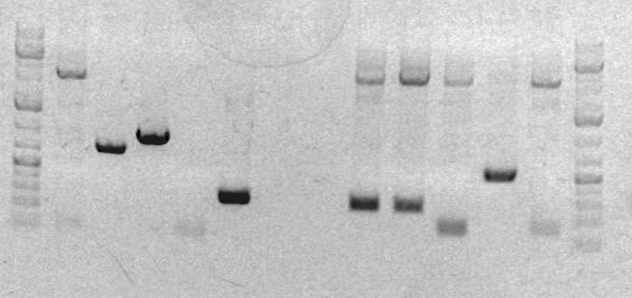
- Results of the analytical digest showed that it succeeded. The cells have an approximately full length plasmid: Gel pic
- Ladder
- sbb36 Clone 1
- sbb36 Clone 3
- sbb35 Clone 1 <--
- sbb35 Clone 2 <--
- sbb35 Clone 3 <--
- sbb35 Clone 4 <--
- Ladder
- The next step is to sequence the plasmid to make sure that it is 100% what we wanted and no mutations.
Part sbb35:
Purpose: Create the full correct plasmid (sbb35 part + vector) -- Extract and purify the DNA from the cells
Protocol:
- The cells were miniprepped using protocol: Macherey-Nagel Minipepped
Results
- The next step is to run the DNA on an analytical gel, this will allow us to see roughly whether or not the full sequence is present.
Products labeled:
- sb35 1, sb35 2, sb35 3, sb35 4
3/17
Part sbb20:
Purpose: Create the full correct plasmid (sbb20 part + vector) -- Check sequence of plasmid to target plasmid.
Protocol:
- Sequencing
Results
- Will receive the results later.
Products labeled:
- Took the B6 well for sequencing of the sbb20
Part sbb35:
Purpose: Create the full correct plasmid (sbb35 part + vector) -- Determine if the full sequence is present.
Protocol:
- Ran an analytical digest map using protocol: Analytical digest mapping
Results
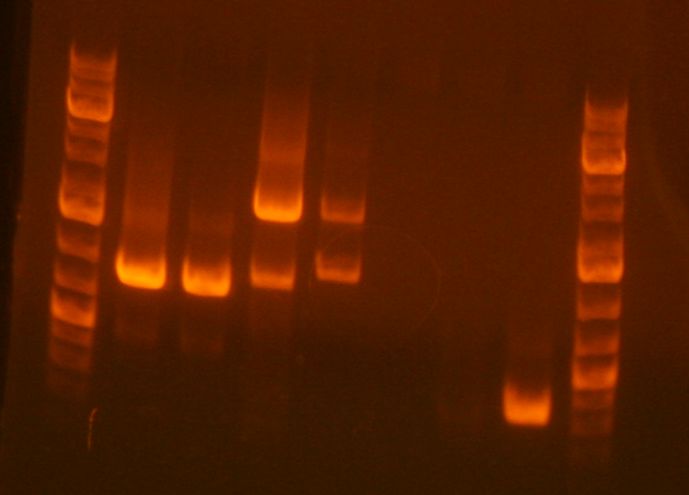
- The result of the analytical digest showed that it failed: Gel pic 1+2 Gel pic 3+4
- Ladder
- sbb07 digest
- JL clone 1 <--
- JL clone 2 <--
- ladder
- JL3 <--
- JL4 <--
- empty
- sbb23-1
- sbb23-3
- sbb23-4
- ladder
- Ran out of time to finish this part and is left as is.