User:Raul M. Cuellar/Growth medium comparison
Investigation topic
Could different types of broth medium yield for higher plasmid concentration in an overnight culture, or does it accelerate cell’s death and reduce plasmid concentration?
Introduction
The performed investigation analyzed the growth rate and final plasmid concentration using three different broth mediums- LB, 2xYT and Super Broth. For this investigation an E. coli DH10B strain carrying pSB1C3 containing aceB insert (BBa_K1119006) was used.
Investigation outline
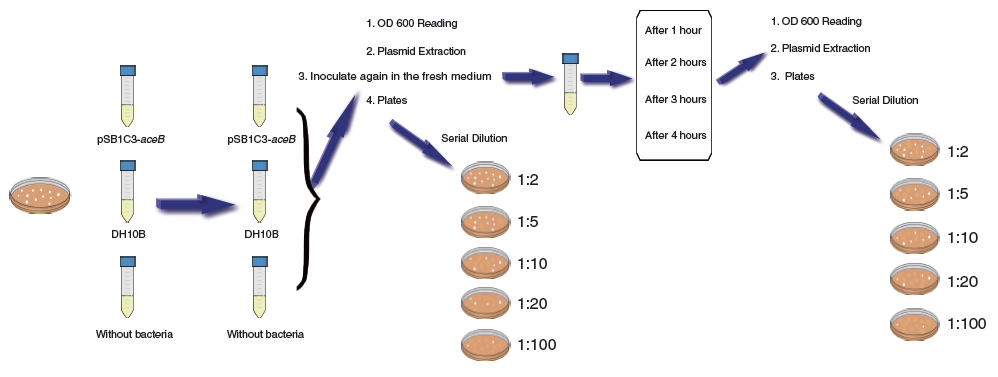
From a given plate of transformed E. coli (DH10B strain) containing pSB1C3-aceB, each self-prepared medium (LB, 2xYT and Super Broth) with added Chloramphenicol (CHL) was inoculated and left to incubate for 16 hours at 37°C (Incubation 1). This is for the purpose of assaying bacterial growth rate and plasmids concentrations.
For assaying bacterial growth rate
Initially 1mL samples from Incubation 1 were taken to make OD readings using a spectrophotometer at 600 nm, using the corresponding pure medium as a blank.
Secondly, samples would be taken from each culture (Incubation 1) for re-inoculation in fresh mediums, giving Incubation 2. This establishes an estimated known initial amount of cells to be cultured, allowing us to determine the growth rate. Samples from Incubation 2 can be assayed for OD changes in time intervals of 1 hr, 2 hrs, 3 hrs and 4 hrs to estimate the bacterial growth rate under the different mediums.
For determination of total cell amount and viable and culturable cells
A sample of each inoculation (Incubation 1) was taken and spreaded over LB-Agar plates containing CHL. This experiment can be used to determine the total viable and culturable cell amount. Serial dilutions were performed for the colony forming units (CFU) quantification in the following factors: 1:2, 1:5, 1:10, 1:20 and 1:100. The CFU can be compared with OD readings (cell total amount) to estimate the concentration of live cells and establish a known cell amount to be incubated to develop Incubation 2 and provide the bacterial growth rate.
For plasmids concentrations assay
Plasmid extraction from the incubated samples coming from Incubation 1 and 2 were made by column extraction using a kit (Favorgen, manufactured in 2009), following the manufacturer's protocol. The plasmid concentration was then measured using a NanoDrop machine readout.
Results
From the methods mentioned, we cultured a DH10B E.coli transformed with pSB1C3 containing aceB insert (BBa_K1119006). After the 16 hour incubation, samples were taken for OD readings. The results can be seen in the following table:

This provided an estimated total bacterial concentration. The growth rate could be estimated using the OD readouts compared with the corresponding CFU observed on the plates.
Due to limitations of the methods employed, serial dilutions were required to be able to estimate the concentration of bacteria. Results from dilutions are as follows:

From the data provided from the OD readouts, we could observe bacterial growth in the inoculated samples. The results for the negative controls were near zero, suggesting no bacterial growths.
To provide an end point, the sample was re-inoculated into a fresh medium and the OD readings and CFU on plates were to be taken once again.
Samples of Incubation 1 were inoculated onto agar plates (in serial dilutions of 1:2, 1:5, 1:10, 1:20 and 1:100) to determine the CFU. However, after a 16 hour incubation at 37°C, bacterial colonies on the experimental plates were too dense to be counted, even for the plate inoculated by a factor of 1:100. Furthermore, bacterial growths were clearly observable in a significant number of the negative control plates. This suggested bacterial contamination. Consistent in every negative control, therefore CFU count would then be unreliable, so the amount of live cells could not be determined.
Due to time constraints, a re-inoculation of plates and overnight inoculations were not performed, and for further confirmation the overnight inoculations were inspected with the discovery of a dense bacterial growth in the negative controls. Centrifugation of the negative control cultures resulted in a significant amount of cell pellet.
Plasmid extraction was carried out from samples containing pSB1C3-aceB from Incubation 1 for restriction check for the presence of the transformed plasmid. The DNA concentration of the product obtained from the plasmid extraction was measured using a NanoDrop:

The low plasmid concentrations indicated that the bacteria cultures contained a low number of the pSB1C3-aceB plasmid.
Conclusion
From both the OD readings and the high number of colonies on the plates, including negative control plates; it is suggested that cross contamination of other bacterial strains occurred since the plasmid count was low. The high concentration of cells observed from the OD readings and plates could have been from bacterial strains that do not contain the pSB1C3-aceB plasmid which were not intended to be cultured.
Materials
- NanoDrop machine: Thermo Scientific NanoDrop 2000 Spectrophotometer
- Escherichia Coli DH10B strain obtained from the KC lab
- pSB1C3-aceB obtained from Hong_Kong_UST 2013 iGEM team glycerol stocks, transformed bacterial done by Chloe Hyun-jung Lee