User:Megan L. Channell/Notebook/Horseradish/2013/09/03
 Biomaterials Design Lab Biomaterials Design Lab
|
|
Adenosine and Inosine UV-VisObjectiveFinding the molar absorptivities of adenosine and inosine. This will help later on in studying adenosine deaminase (ADA) which converts adenosine to inosine.. The structural differences between the two molecules is that adenosine has a primary amine while inosine has a carboxy group. ProtocolStock Solutions Two different stocks solutions were made, one adenosine and one inosine
Dilutions A total of 14 dilutions were made
A sample calculation: Amount of Stock Solution.= [10 mL(dilution concentration)/(stock solution concentration)] UV-Vis
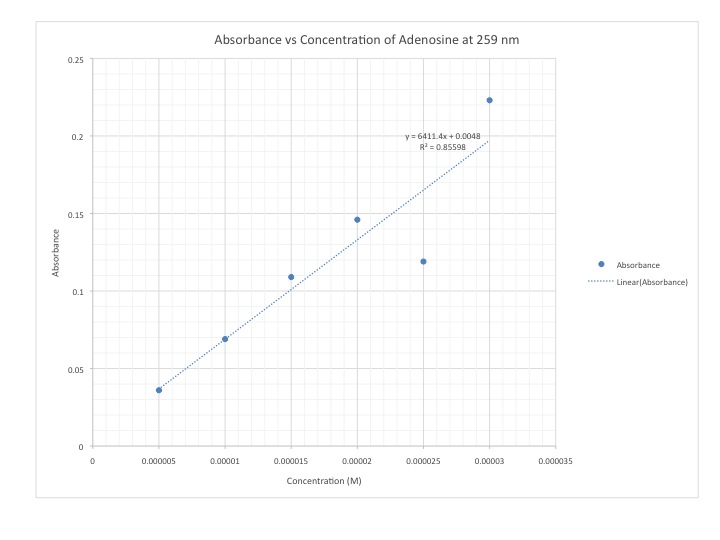
DataA UV-Vis was ran from 800-200nm. The absorbance and calibration curve were done through the data collected on the UV-Vis. The peak of Adenosine was at 259nm and the calibration curve was calculated off this wavelength.
Note
| |