September 10, 2014
Bradford Assay
Prepare Stock Solutions
- Prepare 50 mL of saline solution (0.9 wt-% NaCl)
- 0.9 g→100 mL = 0.9 g/2= 0.45 g NaCl in 50 mL
- Store in 45 mL falcon tube
- Prepare 50 mL of 50 mM Tris base/ 50 mM NaCl solution
- 50 mM →1000 mL 2.5mM → 50 mL
- mass of tris= 2.5 mmol * 121.14 g/mol = 0.303 g Tris
- mass of NaCl= 2.5 mmol * 58.44 g/mol = 0.1461 g NaCl
- Prepare stock solution of BSA 5mg in 5 mL →1.01 μg/μL
- (0.00505 g BSA /mol) * (1 mol/66,430 g) / 0.005 L = 1.5204 X 10-5M = 0.0152 mM
UV-Vis Analysis
Record UV-Vis Spectrum for stock solutions (BSA, Saline Solution, and Tris Buffer)
- Make 7 standard solutions (1mL each) of 1 μg/μL, 4μg/μL, 6μg/μL, 8μg/μL, 12μg/μL, 16μg/μL, and 20μg/mL
- Volume of stock solution is determined based on the concentration chart below.
| Concentration (μg/μL)
|
Amount of Bradford Reagent (μL)
|
Amount of Tris Buffer (μL)
|
| 1
|
200
|
799
|
| 4
|
200
|
796
|
| 6
|
200
|
794
|
| 8
|
200
|
792
|
| 12
|
200
|
788
|
| 16
|
200
|
784
|
| 20
|
200
|
780
|
- All concentrations were diluted to 1 mL total volume with Tris buffer solution and placed in 1.5 mL centrifuge tube.
- Tubes were closed and vortexed for 5-10 seconds and sat for 5 minutes before running UV-VIs.
- Record UV-Vis Spectrum for stock solutions (BSA, Saline Solution, and Tris Buffer) between 200 nm and 800 nm.
- Record UV-Vis Spectrum for 7 standard solutions between 400 nm and 800 nm.
- Make duplicate blanks for
- 1 mL Tris/NaCl buffer
- 200 μL Bradford reagent + 800 μL buffer
- Record their UV-Vis spectra between 400 nm and 800 nm.
NOTE ALL UV-Vis spectra were run using polystyrene cuvettes. Solutions were discarded in waste bottle and polystyrene cuvettes were placed in a wash tube for cleaning.
Data Analysis
1. Purity of BSA stock solution
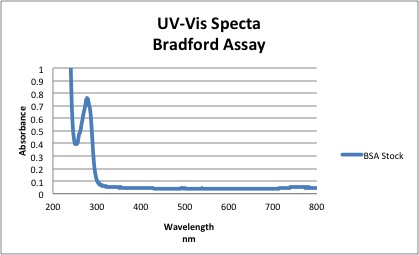
Based on the spectra above, the absorbance of BSA at 280 nm = 0.761
- Using Beer Lambert Law →A= Ε L C
- c=0.761/ 43824 = 1.736 X 10-5M
- Purity= [UV Measured] / [Mass Measured] *100
- 1.736 X 10-5M / 1.5204 X 10-5M *100 = 114%
NOTE The extinction coefficient for BSA at 280 nm was found in literature to be 43,824. The percent purity of BSA was found to be greater than 1 possibly indicating that the polystyrene cuvettes weren't completely clean or BSA did not dissolve completely in solution and stuck to the cuvette. Because the Bradford reagent has peaks at 460 nm and 630 nm and the Bradford-protein complex has a peak near 600, there will be significant overlap. A difference spectra is constructed to make up for the difference and is shown below:

|

