Capillary Electrophoresis - Andrew Maloney, Tim Towner, Camryn Payne
Background
The basic principle of electrophoresis is the movement of charged particles as the result of an electric gradient of roughly 20-30 kV.[1] Electrophoresis is defined by the following equation:
- [math]\displaystyle{ \mu = \frac{Q}{6 \pi r \eta} }[/math]
- [math]\displaystyle{ \mu }[/math] = electrophoretic mobility
- [math]\displaystyle{ Q }[/math] = net charge
- [math]\displaystyle{ r }[/math] = ionic radius of solute
- [math]\displaystyle{ \eta }[/math] = viscosity of solution
Although electrophoresis can easily separate compounds based on differences in charge, it cannot separate compounds where the charge is the same as the ionic radius of the solute or the viscosity of the medium. This situation gives rise to compounds with the same electrophoretic mobility; therefore, they cannot be separated. Capillary electrophoresis further combines the advantages of electrophoresis with capillary flow. The addition of a 50 μm by 100 cm in length creates another key phenomenon, electroosmosis. Electroosmotic flow (EOF) is the phenomenon in which the surface charge on the wall of the capillary interacts with the analyte flowing through by an imposed electric field.[2] The electroosmotic flow is given by:
- [math]\displaystyle{ v=Ε\frac{(\epsilon\zeta)}{(4\pi\eta)} }[/math]
where,
- [math]\displaystyle{ v }[/math] = velocity of the electroosmotic flow
- [math]\displaystyle{ E }[/math] = electric field strength
- [math]\displaystyle{ \epsilon }[/math] = dielectric constant
- [math]\displaystyle{ \zeta }[/math] = zeta potential at the plane of shear near the liquid-solid interface
- [math]\displaystyle{ \eta }[/math] = viscosity of solution
The combination of the electrophoretic flow of charged particles and the electroosmotic flow of the liquid at these high voltages enables a fast separation with well-defined “zones” of the analyte. Essentially, each end of the capillary tube is dipped into a buffer solution and one end is put in the inlet solution with the anode. Meanwhile, the other end runs through the detector to the cathode vial. The analytes will migrate through the capillary due to the electric field gradient and will separate into bands from each other based on size or mobility.
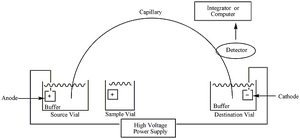
The actual capillary makeup of this technique is critical to separating different compounds. Capillary gel electrophoresis uses gel media for its high surface area and has good separation of analytes based on their size, as caused by the sieving matrix. On the other hand, capillary zone electrophoresis relies on separation based on differences in mobility. Mobility is the key separation factor because the silica capillary is heavily dependent on the characteristics of the buffer solution. Depending on the pH of the buffer solution, different analytes can be separated accordingly with well-defined zones. Also, these zones contain areas of buffer separating zones. As the zones pass the detector they are typically detected with UV absorption near the 200 nm range. Because of the high voltage, and thus tighter bands, the resultant electropherogram will display narrow peaks that correspond to the respective analyte.[3]
Governing Equations
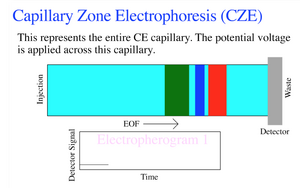
Some of the governing equations for the flow regime in capillary electrophoresis include:[5]
The velocity of an analyte v:
v=μE = μV/L
Where μ is the electrophoretic mobility, V is the voltage, and L is the length of the tube.
Therefore the time for a zone of analyte to transverse the tube t is governed by:
t=L/v or L*L/μV
The molecular diffusion σ that contributes to the broadening of the zones can be modeled by:
σ=2Dt
Where D is the diameter of the capillary and combined with the previous equation, the spatial variance can be modeled by:
σ=2DL*L/μV
A relevant equation for analyzing electropherograms is the resolution which is the distance between peaks divided by the average peak width. [6]
Transition to the Microscale
In the case of capillary electrophoresis, there are many benefits of transitioning to the microscale including the ability to apply high voltages because of the heat transfer on the small diameter of the capillary. [7] This high voltage is what yields the strong zone definition and allows the process to be run relatively quickly. The small diameter allows for heat to be generated uniformly throughout the tube's cross-section and dissipated to the tube walls.[1]
Important factors to consider at the microscale are the analytes' viscosity, pH, and polarity. These all affect the flow within the capillary. The viscosity of the analyte and solution changes the flow within the capillary, as seen in the electrophoretic mobility equation and the electroosmotic flow equation. The viscosity of the solution will ultimately slow the rate of the electrophoretic mobility and the electroosmotic flow. Altering the pH of the solution is one of the most popular methods for controlling the rate of the solution.[8] For silica capillaries, the silica's isoelectric point is easily changed depending on the pH of the solution. The capillary contains an intrinsic negative charge; therefore, creating a double layer of electrical charge at the surface. Positively charged ions counteract the net negative charge of the capillary as one layer while the other contains ions that have not diffused toward the capillary. When the pH is changed, the forces controlling the flow rate change. In basic conditions, the electroosmotic flow is stronger than the effects of the electrophoretic migration. While in acidic conditions, the electrophoretic forces are stronger than the electroosmotic ones. Finally, the polarity of the analyte can change the flow inside the capillary. If the polarity of the analyte is strong, this can directly interact with the negative charge on the silica capillary. Further, this interaction decreases the electrophoretic and electroosmotic flow in the device due to the competition between the analyte and the solution. The high polarity can cause strong interactions between the capillary walls or be bound to its solvent so strongly that it cannot flow. In contrast, if the analyte is more hydrophobic, the solution is less likely to interact with the capillary; thus, increasing the flow rate. Additionally, the concentration may affect the conductive properties of the medium; therefore, a low concentration of analytes in the electrolyte buffer to avoid sample overloading. The lower concentration and the smaller volume also place a higher priority on greater levels of detection sensitivity. [1]
Methods of Detection
Once the analytes can be separated from the electrophoresis, it is imperative to detect them. There are many different methods for detecting analytes in capillary electrophoresis including, mass spectrometry, optical spectroscopy, and others. [9] Mass spectroscopy is one of the most popular analytical techniques used in characterizing the differences in analytes. Mass spectrometry paired with capillary electrophoresis or CE-MS can create highly sensitive, chemical information about the analytes. CE-MS works once the analytes have been separated based on the difference in electrophoretic or EOF mobilities in the capillary and the mass spectrometer will detect the difference.
Optical spectroscopy for detection methods primarily consists of fluorescence microscopy and UV absorption for capillary electrophoresis. Using fluorescence as a method of detecting information from capillary electrophoresis, analytes can be excited by a beam of light. Depending on the chemical structures of the different compounds in the analytes, they will fluoresce differently. [9] UV absorption of compounds as a detection method for capillary electrophoresis works similarly to fluorescence but in the opposite way. UV absorption with capillary electrophoresis works by shining light on the column and determining which compounds absorb the light. The difference in which the compounds absorb different wavelengths of light is directly related to the difference in chemical compounds located inside the analyte.
Applications
Capillary electrophoresis continues to be a very highly sensitive technique capable of analyzing a wide variety of compounds. As of recent, the primary applications for capillary electrophoresis consist of the analysis of proteomics and amino acids.[4] Combined with some of the detection techniques mentioned before, amino acids can easily be detected. Depending on the type of amino acids, the ones containing chromophores can easily absorb UV. If the analyte contained amino acids consisting of aromatic compounds and nonaromatic compounds, there would be a noticeable absorption of UV for the compounds with aromaticity present in their structure. This relationship and the high voltage applied to the system create a strong separation technique by providing very narrow and well-defined peaks. Although amino acids can be detected with these techniques, not all amino acids are UV-active. Capillary electrophoresis can also use the buffer as a way of separating amino acids based on their pH in a solution. [4] Rainelli et al. was able to differentiate a variety of amino acids, like threonine and methionine, based on their pKa in the buffer solution.
Capillary zone electrophoresis can also be applied to separate larger things like proteins. For a proper run of a capillary zone electrophoresis separation with proteins, the proper pH level needs to be maintained via the buffer to overcome the Coulombic repulsion of the protein and the silica walls. [10] This technique also offers a low-cost and user-friendly way to analyze the quality of antibiotics such as penicillin. Paul et al. describe a method for analyzing the quality of pre or post-commercialized antibiotics using capillary zone electrophoresis. [7]
References
1. Jorgenson, J. W. Capillary Zone Electrophoresis. New Directions in Electrophoretic Methods 1987, Chapter 13, pp 182-198. https://doi.org/10.1021/bk-1987-0335.ch013.
2. Alizadeh, A.; Hsu, W.-L.; Wang, M.; Daiguji, H. Electroosmotic Flow: From Microfluidics to Nanofluidics. Electrophoresis 2021, 42 (7–8), 834–868. https://doi.org/10.1002/elps.202000313.
3. Lele, M.; Lele, S. M.; Petersen, J. R.; Mohammad, A. Capillary Electrophoresis. In Clinical and Forensic Applications of Capillary Electrophoresis; Petersen, J. R., Mohammad, A. A., Eds.; Humana Press: Totowa, NJ, 2001; pp 3–19. https://doi.org/10.1007/978-1-59259-120-6_1.
4. Cheng, Y.-F.; Dovichi, N. J. Subattomole Amino Acid Analysis by Capillary Zone Electrophoresis and Laser-Induced Fluorescence. Science 1988, 242 (4878), 562–564. https://doi.org/10.1126/science.3140381.
5. Jorgenson, J. W.; Lukacs, K. DeArman. Zone Electrophoresis in Open-Tubular Glass Capillaries. Anal. Chem. 1981, 53 (8), 1298–1302. https://doi.org/10.1021/ac00231a037.
6. Paul, P.; Sänger-van de Griend, C.; Adams, E.; Van Schepdael, A. A Simple, Low-Cost and Robust Capillary Zone Electrophoresis Method with Capacitively Coupled Contactless Conductivity Detection for the Routine Determination of Four Selected Penicillins in Money-Constrained Laboratories. Electrophoresis 2018, 39 (20), 2521–2529. https://doi.org/10.1002/elps.201800033.
7. Jorgenson, J. W.; Lukacs, K. D. Capillary Zone Electrophoresis. J. Chromatogr. Libr. 1985, 30, 121–131. https://doi.org/10.1016/S0301-4770(08)60829-5.
8. Melanson, J. E.; Baryla, N. E.; Lucy, C. A. Dynamic Capillary Coatings for Electroosmotic Flow Control in Capillary Electrophoresis. TrAC Trends in Analytical Chemistry 2001, 20 (6), 365–374. https://doi.org/10.1016/S0165-9936(01)00067-X.
9. Voeten, R. L. C.; Ventouri, I. K.; Haselberg, R.; Somsen, G. W. Capillary Electrophoresis: Trends and Recent Advances. Anal. Chem. 2018, 90 (3), 1464–1481. https://doi.org/10.1021/acs.analchem.8b00015.
10. Lauer, H. H.; McManigill, D. Capillary zone electrophoresis of proteins in untreated fused silica tubing. Anal. Chem. 1986, 58 (1) 166–170. https://doi.org/10.1021/ac00292a041
