Shlo/notebook: Difference between revisions
No edit summary |
No edit summary |
||
| Line 1: | Line 1: | ||
[[Media:lux_overnight_fluorescence_7_11_07.xls]] | |||
<b> Helpful Results/Links</b> | <b> Helpful Results/Links</b> | ||
Revision as of 07:31, 12 July 2007
Media:lux_overnight_fluorescence_7_11_07.xls
Helpful Results/Links
Growth Curves
Plate Reader Protocol
Week 4
7/12
I miniprepped some liquid cultures that were grown last night:
- R0040
- R0011
- P0140(-1)
- P0340(-1)
- R0051
7/11
Note: We had set up some fluorescent readings on the plate reader to run for a few hours yesterday, but somehow the PC restarted and failed to save/restore our information. Other than a few observations that we had made during the process - such as T9002 (+) growing much faster than T9002 (-), all our data was lost ... very, very unfortunate and aggrevating.
Plans
- Figure out J-T: start with low concentration, see if fluorescence spikes (which might indicate quorum-like activity) rather than slow growth of fluorescence.
- Re-Colony PCR some of the constructs (list in my notebook)
- New growth curves?
Results from FACS
All inductions were at 2 hours with 100nM OHHL.
We've also learned that we should do about a 1:50 dilution of the overnight cultures. 0.03 OD is probably optimal.
- T9002 (non-induced) had some leaky fluorescence.
- T9002 (induced) showed strong fluorescence as expected.
- I73 (non-induced) had a little less leaky fluorescence than T9002, but still had some leakiness.
- I73 (induced) showed strong fluorescence as expected. S08-I07 + T9002 had little fluorescence in the GFP but had lots of fluorescence for RFP.
- S08-I07 + I73 had strong fluorescence for both YFP and RFP. It should be noted, though, that the count of YFP was around 500 at peak, while some of the induced were around 1250 at peak. Perhaps this has to do with inconsistent cell numbers?
- S08 I07-T9002 had very little GFP (low count, both negative and positive) but strong RFP.
- J39-T9002 showed strong GFP fluorescence.
- 15311 showed strong YFP fluorescence as expected, with peak counts around 1600.
- I13522 showed strong GFP, as expected, with peak around 1750.
- B0015 served as the negative control
- F2620/E40 (induced) showed no fluorescence, nor did the F2620/E40 (non-induced). Probably time to let go of the F2620.
Setup for FACS
ODs and fluorescence taken from the plate reader to figure out dilution and efficacy of some of the overnight dilutions/combinations.
Induction, 100nM final concentration OHHL
| Sample | OD | Fluorescence (excitation485, em38) | OD after one hour of growth, at induction |
| T9002 (+) | 1.486 | 675.85 | not used |
| T9002 (-) | 1.212 | 212.61 | 0.379 |
| I13273 | 1.283 | 208.69 | 0.430 |
| F2620-E0240 | 1.487 | 191.98 | 0.527 |
| J23039+T9002 | 1.174 | 4698.8 | 0.317 |
| B0015 | 1.470 | 174 | 0.496 |
| I13522 | 1.571 | 7978.7 | 0.397 |
| I5311 | 1.643 | 2258 | 0.482 |
| S08-I07 w/T9002 | 1.313 | 2538 | 0.427 |
| S08-I07 w/ I73 | 1.248 | 1701 | 0.395 |
Interestingly, the T9002 (+) had relatively little fluorescence compared to the constitutve samples. Perhaps the fluorescence had started to decrease over time, or perhaps the T9002 was not activated that strongly ... this is something we can measure over time with the plate reader ... if it does not turn off again and lose our data.
8mL LB + Amp (except for I5311, with Kan) added to enough uL of cells to make a "cell mass" of 371.5.
Setup for Overnight Plate Reader
A1-3: T9002 (noninduced)
A4-6: T9002 (+100nM OHHL)
B1-3: I13273 (noninduced)
B4-6: I13273 (+100nM OHHL)
C1-3: F2620-E0240 (noninduced)
C4-6: F2620-E0240 (+100nM OHHL)
D1-3: J39-T9002
D4-6: B0015
E1-3: I13522
E4-6: I5211
F1-3: S08-I07 w/T9002
F4-6: S08-I07 1/ I73
7/10
Plate Reader
Prepared plate reader for fluorescence with OHHL.
A1-3: T9002 (-), 10nM OHHL
A4-6: T9002 (+), 10nM
B1-3: T9002 (-), 100nM
B4-6: T9002 (+), 100nM
C1-3: I13273, 10nM
C4-6: I13263, 10nM
D1-3: I13273, 100nM
D4-6: I13263, 100nM
E1: B0015, negative control
E2: T9002(-), no induction
E3: T9002(+), no induction
E4: I13273, no induction
E5: I13263, no induction
E6: I13522, no induction (constitutive GFP => positive control)
Note: T9002 (+) indicates that the T9002 sample was incubated with 1000nM OHHL.
The samples were all put in 1:20 (sample:total volume), total volume = 200uL, inclubes LB + Amp.
Induction expected to occur around 1hr20min after dilution, at 0.3OD, around 12:15.
Colony PCRs
Prepared 3 different colony PCRs & E-gels.
Extension for 1min15sec
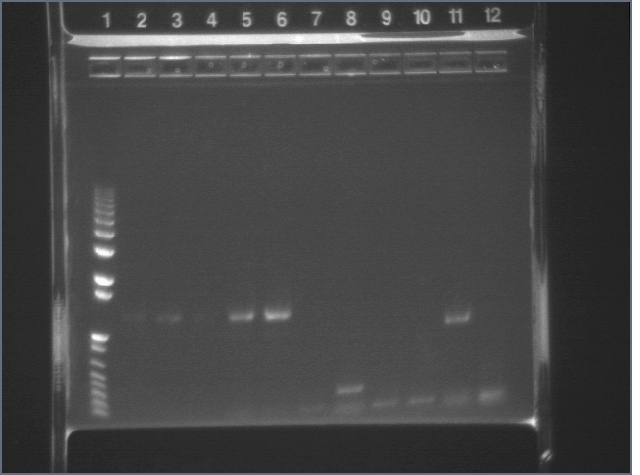
Lanes 2-6: F2620
Lanes 7-12: B0015
Extension for 2min15sec

- Ladder
- I15030 1
- I15030 2
- F26-E 1
- F26-E 2
- F26-E 3
- F26-I13507 1
- F26-I13507 2
- F26-I13507 3
- S08-E0240 1
- S08-E0240 2
- S08-E0240 3
- Ladder
- J37015 1
- J37015 2
- J37015 3
- J37015 4
- J37015 5
- J39-T9002 1
- J-T 2
- J-T 3
- J-T 4
- J-T 5
- J-T 6
Readings: SdiA and AHL
Interestingly, on our plate reader today, T9002(+) samples grew consistently faster than the T9002 (-) and other samples. The I63 and I73 samples grew at the same rate despite being in different types of cells (Top 10 vs BL21).
Some papers seem to suggest that SdiA acts similarly to a LuxR and is involved in cell division processes (though papers also mention that exact mechanisms remain unknown). One paper has found that addition of AHL to e coli should not affect their overall growth, though a number of genes are upregulated and downregulated, including OmpC (upregulated). [[1]]
Additional Readings:
- [Control of cell division in Escherichia coli: regulation of transcription of ftsQA involves both rpoS and SdiA-mediated autoinduction.]
- [Effect of sdiA on Biosensors of N-Acylhomoserine Lactones]
7/9
Perry, George, and I did a number of colony PCRs on various samples. Results below. The ones that worked are marked.

- Lane 1: Ladder
- 2: J37015, 1
- 3: J37015, 2
- 4: J06702, 1 (as expected)
- 5: J06702, 2
- 6: I6042, 1 (as expected)
- 7: I6042, 2 (as expected)
- 8: I13273, 1
- 9: I13273, 2 (as expected)
- 10: F2622, 1 (as expected)
- 11: F2622, 2
- 12: H2O
- Lane 1: Ladder
- 2: F26-I07
- 3: S08-E40
- 4: S23-I07 (as expected)
- 5: S23-E0240 (as expected)
- 6: F26-E40
- 7: S08-I07 (as expected)
- 8: F2621, 1 (as expected)
- 9: F2621, 2 (as expected)
- 10: I15030, 1
- 11: I15030, 2
- 12: H2O
Made liquid cultures of a number of samples for plate reader tomorrow.
Also, left one portion of T9002 incubating with about 1000 nM OHHL to serve as comparison/control of fluorescence, to J37015.
Week 3
07/06
Helped Perry prepare a colony PCR of F2620 and a few other samples
Planning to do some transformations at the end of the day, including some of the parts we found: J06702, F2622, F2621, I15030 (we should have this somewhere), !6042, I13273, J37015 (?)
Found Imperial's page from last year, with testing of the T9002 part we're using. Interestingly, as they varied AHL concentration, fluorescence did not seem to change too significantly. Imperial T9002 Results
07/05
Prepared a colony PCR using some of the plates that I moved yesterday.
The PCR looks really bad/has hardly any bands, which is really confusing. We might have to redo this all ...
Additional Readings and Thoughts
Trying to read to trigger brainstorming ideas for fluorescence assay as well as gauge what to expect.
Harris poses a good question: why do we have luxR/luxI constructs in different bacteria? Will probably be a lot less sensitive than if we had both in one ... though perhaps the applications of having a sender/receiver are more diverse? Time to discuss..
Again, the Andersen et al paper (gfp-based N-acyl homoserine lactone sensor systems for detection ...) is extremely helpful in this regard:
- Their construct (e. coli MT102) was much more sensitive to OHHL relative to other proteins in the acyl-homoserine lactone family.
- 1 nM OHHL concentrations did not cause fluorescence to be any higher than background. Significant fluorescence began at about 3nM OHHL. 100nM OHHL was only slightly higher in fluorescence compared to 10nM OHHL; as I've noted before, "maximal induction" was coined to be at 10nM OHHL.
- GFP: excitation of 475nm and emission at 515nm
- Interestingly, the fluorescence peaked around 250 minutes, and then started to decrease (... this could do with their different GFP constructs, though).
Ripp et al: Linking bacteriophage infection to quorum sensing signalling and bioluminescent bioreporter monitoring for direct detection of bacterial agents
- We've been talking about trying to measure amounts of OHHL produced, so we can detect whether our lux I construct is working. n this paper, OHHL concentrations were analytically determined using liquid chromatograph-mass spec. At least it's feasible, though I don't think our lab could/would do this.
- Induced E. coli OHHLux bioreporter using synthetic OHHL.
- "significant" signals were considered to be those 2 standard deviations above background.
- Saturation-type behavior was observed at OHHL concentrations exceeding 50uL ... which is much higher than anything we've used
- Found a linear-type response at OHHL concentrations from 20nM to 2uM
Burmolle M et al: Presence of N-acyl homoserine lactones in soil detected by a whole-cell biosensor and flow cytometry
- To figure out dose-response, OHL and samples shook at 37C for 20 hours (including antibiotic and LB, 5mL total). Afterward, 1mL was washed once in PBS (for each sample) and resuspended in 3mL PBS for fluorescence readings
A bunch of papers also seem to reference TLC (thin layer chromo) as a possibility to detect/characterize OHHL levels.
07/04
Took plates out of incubator, put into fridge
F2620, top 10: lawns of colonies
JL0159: lawns
S03623-B0015: a few
JL0159-PDZ1+2: minimal growth
JL0159-PDZ2: some growth
S03608-B0015 in Top 10... are these air bubbles or colonies?
JL0159-PDZ1: Minimal growth
I put the E0240 in the freezer. Be careful; the non-digested microcentrifuge tube is there as well!! The digested one is clearly labeled "digested".
shlo/notebook/0704fluorescence
07/03
shlo/notebook/growthcurves0703 ... Growth curves for BL-21 with the lux insertions. Most of them look pretty good; should serve as a general reference for all groups, until (if?) you make your own growth curves.
Helped Perry with some ligations/plating, and started a 5-hour fluorescent/induction experiment. First, I loaded the machine with cells (dilution - 1:10 ... maybe will dilute in greater amounts next time, eg 1:100?) and watched OD. Around 0.3OD I took them out, transferred to a clear/black 96-well plate (next time, will start with one of these), and induced cells with OHHL.
Wells A1-3: I5311 (constitutive YFP, kan)
Wells A4-6: I13522 (constitutive GFP, amp)
Wells B1-3: BL-21 with PET29 OmpA1+His (negative control, kan)
Wells B4-6: BL-21 with I13263 (negative control, inducible for YFP, amp)
Wells C1-3: BL-21 with I13263 (10nM OHHL added)
Wells C4-6: Top10 with T9002 (10nM OHHL added)
Wells D1-3: BL-21 with I13263 (100nM OHHL added)
Wells D4-6: Top10 with T9002 (100nM OHHL added)
Also, digested some E0240 and plated S03608 and S03623, along with B0015. We'll use this to re-create the J37034 that hasn't seemed right (though our own constructs will have different promoters ... which is good in the long run). All of these samples (plates and microcentrifuge tube) are in the incubator, and someone will have to move the plates to the fridge and the tube to the freezer tomorrow.
07/02
Nanodropped some of the samples from yesterday and Saturday before Perry digested them and we sent them out for sequencing.
| Sample | Nanodrop result (ng/uL) |
| S03623, Sample 1 | 104.5 |
| S03623, Sample 2 | 111.2 |
| S03608, Sample 1 | 126.7 |
| S03608, Sample 2 | 110.3 |
| J37034, Sample 1 | 196.5 |
| J37034, Sample 2 | 97.6 |
| J23039 | 65.8 |
| R0011 | 61.4 |
| R0140 | 86.1 |
| T9002 | 185.1 |
| B0015 | 71.6 |
| R0051 | 100.2 |
We wanted to construct a growth curve for our cells, so we took the refrigerated BL21+I13263 cells (from Friday) and made 800uL of different dilutions for the plate reader. Ampicillin and LB were added to each sample, and samples were made for 1:10 (wells A1-3), 1:20 (B1-3), 1:30 (C1-3), 1:40 (D1-3), 1:50 (E1-3), 1:60 (F1-3), 1:70 (G1-3), 1:80 (H1-3), 1:90 (A4-6), 1:100 (B4-6), and of the BL21 cells (1:10, C4).
Also, diluted the OHHL to 0.05M. There is 938uL total.
Grew liquid cultures of I5311, OmpA1+his, I13522, T9002, and I13263. These will be used when we induce tomorrow and use the Plate Reader to detect fluorescence.

See Perry's notebook for more info on this.
Week 2
7/01
Did another miniprep of some Biobricks that Perry had grown in liquid culture. The tubes are in the freezer at my workstation. Perry helped me visualize the Egel from yesterday (the machine is somewhat broken ... I had left the gel in the fridge overnight. Though some of the gel diffused, the bands are still distinguishable). The samples were known to be either R0051, B0015, or T9002. Band 3 ran the slowest, indicating the highest molecular weight, and therefore T9002. B0015 is 129 bp whereas R0011 is 55bp, so sample 2 (which ran the furthest) was likely to be R0011, and sample 1 was B0015.
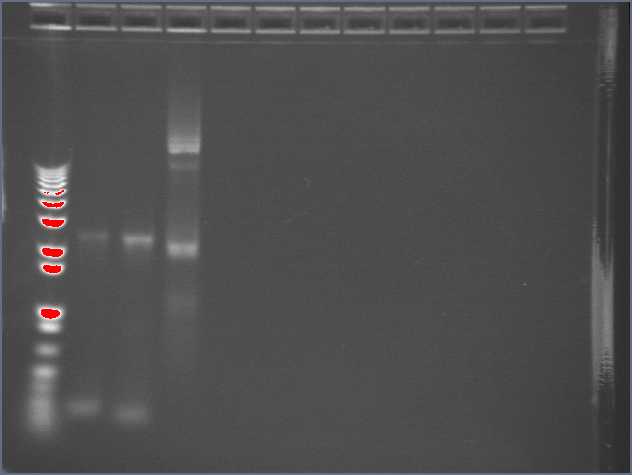
6/30
Helped miniprep some Biobricks, but 3 of the labels smeared and I was unable to discern them. PCR'd them, Will run an E-gel.
E-gel:
Lane 1: 1kb+ ladder
Lane 2: Sample 1
Lane 3: Sample 2
Lane 4: Sample 3
Rest of lanes loaded with water as instructions dictate.
6/29
... oh lord. i have so much more to add. sorry for the delay guys ... mad busy day.
George, Perry, and I arrived at 8:15am to start to prepare the bacteria for the FACS appointment at 3:30. We took the optical densities of the cells we grew in liquid medium overnight and got 1.785, 1.793, 1.780.
9am: We prepared 3 tubes to 2mL each, with dilutions of 1:40, 1:80, 1:100. 2uL of Amp was added in each, and the following:
1:40 - 50uL cells, 1498uL LB
1:80 - 25uL cells, 1748uL LB
1:100- 20uL cells, 1792uL LB
9:15am: Made and set a 1% agarose gel for the digest we prepared yesterday.
Also helped Perry with some of his colony-PCRs. Gel has 3 lanes- 1: ladder, 2: yesterday's digest, 3: Perry's sample. Will be visualized using SyberGold
10:30am: OD of 4-hr samples. Realized we had not diluted them enough. Diluted 1:100 for the spectrometer (so we did not use up too many samples), had reading of 0.03 for 1:40, 0.018 for 1:80, 0.013 for 1:100. Decided to re-dilute 1:100 for samples originally marked 1:40 and 1:100, and 1:30 for sample originally 1:80. Gives us an absolute OD of 0.03 for the 1:40, 0.01 for the 1:100, and 0.05 for the 1:80. (made 2mL total for each sample, adding in cells, LB, and Amp)
1:40- 20uL cells, 2mL LB, 2uL Amp, 1:80 - 65uL cells, 2mL LB, 2uL Amp, 1:100 - 20uL cells, 2mL LB, 2uL Amp.
Also: prepared 2hr samples
11am: George prepares HSL solution using 200proof ethanol.
11:30am: Prepare the 1-hr incubations
12 noon: OD of 4-hour induction, right before protein added. 1:100 dilution done for spectrometer. (10uL cells, 990LB). The numbers below are the raw numbers from the machine - so our samples actually have OD 100x this.
1:40 - 0.002
1:80 - 0.002
1:100 - 0.001
Since we didn't have as much volume of cells as we had anticipated, instead of doing 2mL cell samples as planned, we used 1mL of cells with 10.5 uL HSL (diluted by George - see his personal notebook page). 2 samples were taken from the 1:40 first dilution (8:30am) - used for the 10 and 100nM HSL runs - and 1 from the 1:80, with 1nM HSL.
1:30pm: Induced the two-hour samples. First, OD taken of 2-hr and 1-hr (1hr had been growing for about 1.5 hours at this point).
Again, all ODs diluted 1:100 first.
Two hours: 1:40 - 0.078, 1:80 - 0.035, 1:100 - 0.005. One hour: 1:40 - 0.016, 1:80 - 0.008, 1:100 - 0.007. Also, we had left the 4-hour cells on the bench (from before) - ODs of 0.002 (1:80) and 0.004 (1:100).
2 samples from the 2-hour 1:100 (0.005 OD) were used to make the 10, 100nM HSL.
Since the ODs were already so high for the 1-hr, we also decided to take our original overnight liquid cultures and dilute them for the 1-hr. We used the same formula as above and diluted to 1:80 and 1:100.
2:30pm: 1-hr rediluted samples gave 0.003OD (1:80) and 0.002 (1:100). We used the 1:80 for the 1nM HSL and the 1:100 for the 10, 100nM HSL.
A negative control was also created from 1mL of the 1:80 solution.
3:15pm: All samples spun down for 2 minutes at 10,000rpm, supernatant removed, reconstituted with 0.5mL PBS (1x)
Colony counts from 06/28:
275uL
OmpA1 + 10mer: 167
OmpA1 + 15mer: 266
OmpA1 + 20mer: 224
OmpA2 + 10mer: 60
OmpA2 + 15mer: 9
OmpA2 + 20mer: 28
10uL
OmpA1 + 10mer: 15
OmpA1 + 15mer: 8
OmpA1 + 20mer: 0
OmpA2 + 10mer: 1
OmpA2 + 15mer: 0
OmpA2 + 20mer: 0
6/28
George, Perry, and I transformed some of the BioBricks into 20uL of Top Ten cells.
We want to change the pL promoter because it does not allow for constitutive expression. Some of the Biobrick parts we're using may facilitate this.
Also: grew cells in liquid media, digested BB, looked at sequences and discovered that the BB isn't what it should be - should be about 800bp, with ribosome binding region and terminator region, but was only about 300bp, with terminator and something following it.
6/27
Yesterday night I left some vectors & plasmids to ligate in the 37C incubator. Kevin took the vectors for nucleotide removal, and I speed Vac'd the plasmids. Originally I was going to make a gel and run the plasmids on multiple lanes for gel extraction, but given the large Nanodrop values (242 - 371 ng/uL) and the large amount of Midiprep used (50uL), Bill suggested that I PCR purify them instead, which gives 95% recovery rather than 80% recovery. A gel will still need to be run to confirm that the samples were indeed ligated.
The plasmids are Lpp+OmpA1+pet29 and Lpp+OmpA2+pet29.
- Reconstitution into 30uL of H2O per plasmid (two plasmids total - OmpA1, OmpA2),
- split into two samples each (so 15uL total volume)
- PCR purified (PCR purification kit from Qiagen)
- Eluted with 50uL nuclease-free H2O per tube
- prepared a gel using TBE
- prepared 5 lanes
- Lane 1: 10uL ladder (1kB +)
- Lane 2: 1uL OmpA1 plasmid (diluted to 20uL total using H2O)
- Lane 3: 3uL OmpA1 plasmid (diluted to 20)
- Lane 4: 1uL OmpA2 plasmid (diluted to 20)
- Lane 5: 3uL OmpA2 plasmid (diluted to 20)
The gel was run at 130V for 45 minutes.
4 tubes total should remain: 2 tubes of each type of plasmid. 2 tubes should have 49uL left (labeled "1"), 2 tubes should have 47uL left (labeled "2"). After gel ran for 45 minutes, bands appeared about 3/4 way down the gel. Ethidium bromide (2uL) and 100mL TBE buffer used, put on shaker for 45 minutes. Visualized under Trans-UV.

Nanodrop, 6/27:
10mer: 122.3 ng/uL
15mer: 156.3 ng/uL
20mer: 167.4 ng/uL
plasmidOmpA1: 39.6 ng/uL
plasmidOmpA2: 42.8 ng/uL
We Speed Vac'd the plasmids and resuspended both in 33uL of nuclease-free H2O. To calculate volumes of sample needed , we approximated that the vector was 5800 in size and the insert was 50. Therefore, we'd need 1200ng of vector and 103ng of insert.
Protocol for Ligation of Plasmid given Nanodrop results and volumes available
OmpA1 or A2: 10uL
10mer, 15mer, or 20mer: 1uL
buffer: 1.5uL
ligase: 1uL
dH2O: 1.5uL
(total of 15uL)
Leave in PCR machine (left in the one at George's workstation) overnight for 15C. The extra 10,15,and20mer tubes were put in the "Mike Strong iGEM plasmid" box in the freezer in the small laboratory. We used shorthand for the small PCR tubes: 110 means OmpA1+10mer, 115 means OmpA1+15mer, 210 means OmpA2+10mer, etc.
Readings and Brainstorming
Idea: increasing ligation efficiency
According to a paper I just read, (Lund et al) "temperature cycle ligations" may provide a 4-8 fold increase on cloning efficiency, since the cycling balances high enzyme activity and DNA annealing. The temperature cycle should run for 12-16 hours, cycling between 30 second bouts at 10C and 30C.
Readings toward QS and lux
Bacterial Transformation Experiment James Slock has a detailed protocol for transforming the lux genes into e.coli. Actually, the transformation protocol is the same as what we've been doing, but there is some specific info on lux stuff. Not really essential, I guess.
MIT Parts Registry: Overview of LuxR system
[ww.ai.mit.edu/projects/ cellular-robotics/rweiss-dna6.ps Paper] Really nice, but needs to be converted to pdf - my Mac did this automatically ...
MIT Parts, BBa_F2620
spatiotemporal control reading Look at the supplementary information as well.
Paper ...some other genes that may be turned on by HSL are mentioned ...
somewhat relevant ... but not completely Seems that the e. coli were grown with OHL... and a different strain was used
BL21 More info on the cells we're using
look in methods for treatment of HSL powder ... mammalian cells, but for method only
shlo/notebook/luxfluorescenceduration
Week 1
Doing some reading on Fec now. Really brief notes and some of the more interesting readings follow.
Fec genes encode proteins essential for ferric citrate transport in e.coliK12.
FecA is an outer membrane protein
=N-proximal 79-residue extension: deletion of this extension abolishes induction by ferric citrate but retains feric citrate transport: Kim et al, Transcription induction of the ferric citrate transport genes via the N-terminus of the FecA outer membrane protein, the Ton system, and the electrochemical potential of the cytoplasmic membrane Kim article
Gene regulation by transmembrane signaling Some really nice info on structure of Fec and interactions with ferric citrate
Biocyc
- Nakajima H, Shimbara N, Shimonishi Y, Mimori T, Niwa S, and Saya H. Expression of random peptide fused to invasin on bacterial cell surface for selection of cell-targeting peptides. Gene. 2000 Dec 30;260(1-2):121-31. DOI:10.1016/s0378-1119(00)00461-3 |
- Vassylyev DG, Sekine S, Laptenko O, Lee J, Vassylyeva MN, Borukhov S, and Yokoyama S. Crystal structure of a bacterial RNA polymerase holoenzyme at 2.6 A resolution. Nature. 2002 Jun 13;417(6890):712-9. DOI:10.1038/nature752 |
NanoDrop results (6/20) performed by Ellenor and Stephanie:
(1.5 uL used out of a 30uL elution with nuclease free water)
S: 10.9 ng/uL
S2: 15.4 ng/uL
B: 59.1 ng/uL
B1: 25.1 ng/uL
Brainstorming
Brainstorming for the two-component systems (really for my own use for now - not expected to be coherent)
Structural comparison of the PhoB and OmpR DNA binding/transactivation domains and the arrangement of PhoB molecules on the phosphate box
-NMR used to determine 3DE structure of PhoB DNA-binding/transactivation domain. Very similar to OmpR DNA-binding/transactivation domain, except for conformation of the long turn region of PhoB (interaction site for sigma subunit, rather than interaction with alpha subunit for OmpR)
Interdomain linkers of homologous response regulators determine their mechanism of action
Focuses on OmpR and PhoB and, as the title suggests, supports that phosphorylation of sites (particularly N-terminus of both proteins) improves affinity to bind DNA. Isolated C terminus of OmpR is insufficient to productively interact with RNA polymerase.
I've been told by some of the lab members that OmpR is an inner-membrane protein and therefore cannot be used for our assays. It seems that we'll have to find another protein ...
The phosphoryl transfer domain of UhpB interacts with the response regulator UhpA
UhpB = histidine kinase protein that controls production of sugar phosphate transporter UhpT
UhpA = response regulator; when phosphate is transferred from histidine to aspartate, ability of kinase to bind to target DNA sequences and to alter gene transcription is altered.
Major result: indication that phosphoryl, transfer-dimerization of UhpB participates in specific binding of UhpA, in control of autokinase activity, and dephosphorylation of P-UhpA
... So I found this paper on a search on PubMed for e coli outer membrane protein signaling dimerization. However, I've found that UhpA resides in the cytoplasm, whereas UhpB is an inner-membrane protein. Boo.
More thoughts: we could potentially try to target some of these inner-membrane proteins to the outer-membrane. I don't know if this is really feasible - while we can attach the appropriate signal sequence, I'm not sure the environment would allow for correct conformation and activity of said proteins.

