BME103:T930 Group 6 l2: Difference between revisions
| Line 137: | Line 137: | ||
8. Place smartphone with the flash off onto holder and direct camera lens at the slide apparatus. | 8. Place smartphone with the flash off onto holder and direct camera lens at the slide apparatus. | ||
9. Take photo with smartphone and process on computer using ImageJ. | 9. Take photo with smartphone. | ||
and process on computer using ImageJ. | |||
10. Using a clean pipette, remove the sample of diluted water and SYBR green dye from the slide. | 10. Using a clean pipette, remove the sample of diluted water and SYBR green dye from the slide. | ||
11. Repeat steps 4-10 using | 11. Repeat steps 4-10 using included calf thymus DNA as a control, and move back on the glass slide two rows to eliminate contamination. | ||
12. Once there | 12. Once there are no more rows on the glass slide, carefully discard the used slide and begin using a new one. | ||
==Research and Development== | ==Research and Development== | ||
Revision as of 10:48, 27 November 2012
| Home People Lab Write-Up 1 Lab Write-Up 2 Lab Write-Up 3 Course Logistics For Instructors Photos Wiki Editing Help | |||||||||||||||||||||||||||||||||||
OUR TEAMLAB 2 WRITE-UPThermal Cycler EngineeringOur re-design is based upon the Open PCR system originally designed by Josh Perfetto and Tito Jankowski.
Instructions
ProtocolsMaterials
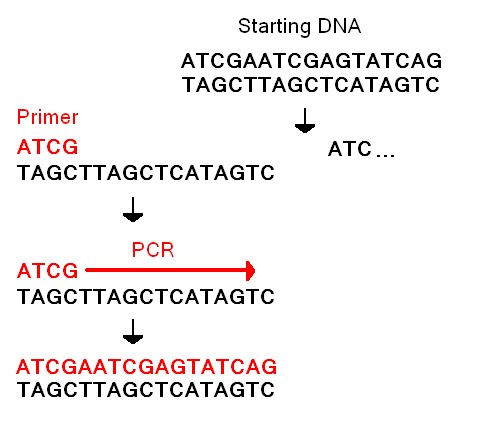
1. Using a micro-pipette, transfer 1.0-1.5 mL of desired DNA sample into at least three test tubes. 2. reagent solution 3. place tubes into holder in PCR machine 4. program cycles on OpenPCR 5. Run reaction
DNA Measurement Protocol 1. Remove lid from black box and invert the box, open the front flap. 2. Place one glass slide into the holder. 3. Depending on the amount of samples being used, take a clean pipette for each sample and label it so that it is only used for that sample of DNA. For example, label a pipette for SYBR green with a horizontal line on the pipette bulb, and label a pipette for diluted water with a vertical line on the bulb. Do NOT use pipettes for any other solution than what they are labelled for. 4. Using the designated pipette, squeeze two drops of SYBR green dye inside the round glass windows on slide, preferably the second dot of the second row. 5. Begin by adding a sample of diluted water to the SYBR green dye, and using the appropriate labelled pipette, add two drops in the same spot as the SYBR green dye. 6. Align the sample so the blue LED shines directly though it focusing the light on the other side. 7. Move holder apparatus inside the box so that no light reaches it. 8. Place smartphone with the flash off onto holder and direct camera lens at the slide apparatus. 9. Take photo with smartphone. and process on computer using ImageJ. 10. Using a clean pipette, remove the sample of diluted water and SYBR green dye from the slide. 11. Repeat steps 4-10 using included calf thymus DNA as a control, and move back on the glass slide two rows to eliminate contamination. 12. Once there are no more rows on the glass slide, carefully discard the used slide and begin using a new one. Research and DevelopmentBackground on Disease Markers
| |||||||||||||||||||||||||||||||||||