User:Dan C. Wilkinson/Notebook/Physics 307L/9/29/10
Electron Diffraction
Equiptment
>Teltron 813 kV power unit (high voltage supply)
>Tel 2501 universal stand
>Tel 2555.02 magnet
>Tel 2555 electron diffraction bulb
>Hewlett Packard 6216B power supply (regulating voltage source)
>connector cables
History
In the early 1900's quantum mechanics was taking form and new, radical ideas were replacing old, intuitive ones. For example the photoelectric effect was only explainable when light was considered to be both a particle and a wave. In 1924 Louis de Broglie made the assertion that if light (previously considered to be a wave) can act like a particle then 'particles' should be able to act like waves. Obviously this line of reasoning was a huge departure from logic and intuition. Though he was proved right when particles like electrons were shown to defract through a crystal lattice, a wave-like property.
Theory
Louis de Broglies equation states that a partcles wavlength is related to its momentum. Also its frequency is related to its energy.
- [math]\displaystyle{ \lambda = \frac{h}{p} }[/math] and [math]\displaystyle{ f = \frac{E}{h} }[/math]
In this experament we will measure the diffraction of an electron beam through a carbon lattice. It can be shown that the lattice spacing is [math]\displaystyle{ d = \frac{4 \pi L h c}{D \sqrt{2 e V m c^2}} }[/math]
Here the accepted values for the d spacing of the carbon lattice are 0.123 and 0.213 nm.
It is also important to note here that the observed diffraction patterns will not be a series of intensity maxima points that one would expect in electron diffraction off different crystal substances. One would be expected to see six maxima points that correspond to the six degrees of symmetry in the crystal lattice of graphite. What is actually observed are 2 maxima rings corresponding to the smallest d-spacings in the crystal structure. These rings are an artifact of how the graphite is formed structurally. Graphite is a series of crystal sheets that have relatively low attraction between one another (this is the property that makes graphite a good writing utensil). This results in subcrystals that are randomly orientated to one another. This random orientation causes intensity rings rather than points because the angle the sheets of crystal make with the beam vary inside the graphite solid. good theory refrence
Curvature of Glass Correction
We measured the diameters of the diffraction pattern rings on a curved glass surface. Therefore it is necessary to account for the curvature of the glass in order to get a diameter that we can use in the above equation for d spacing.
The y distance error can be calculated by.
[math]\displaystyle{ y = R - \sqrt{R^2-\frac{Dobs^2}{4}} }[/math]
It can also be shown that
[math]\displaystyle{ tan(\theta) = \frac\frac{Dobs}{2}{L-y} }[/math]
Finally
[math]\displaystyle{ D=2Ltan(\theta) }[/math]
Procedure
Professor Gold's Lab Manual
The equiptment was hooked up as shown in the diagram and everything was turned on. We were not able to use a volt meter to precisly tune the voltage so we had to rely on what the voltage supply was reading out. I adjusted the voltage supply and Tyler made the measurments of the diffraction ring outer diameters. We made three measurments per voltage and stepped the voltage down by 200 volts every run starting at 5000 volts. We had to adjust the magnet twice in order to keep the center in the middle of the glass so that the curvature correction would hold up.
Data
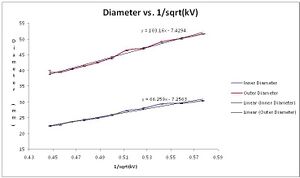
Possible Sources of Error
There are several apparent sources of error in this experiment. The most apparent source is out inability of determine the voltage to more than 2 significant figures. We didn't have a voltage meter that was able to register those high voltages. The next obvious source is the actual act of measuring the diffraction peaks. This measurement is open to human error and human bias. There is also a drift in the center of the patter as the voltage is changed that needs to be corrected by the magnet. This is an important feature to recognize because a drift in the pattern will lead to an incorrect curvature correction. Though all in all we were able to mitigate most of the possible error and get a relatively good estimate of the crystal d-spacing.
Error Correction
Earlier we stated that the measurements we made measured the outer diameters of the diffraction rings. This measurement, while being very precise was flawed. The diffraction pattern 'peak' occurs on the inner diameter of the ring and therefore it is here were the greatest amount of positive interference is happening. To compensate we subtracted off 2mm from each of our measurements (idea from Prof. Koch) and produce results that agree much more with the accepted d-spacing of graphite. These are reported on the above google spreadsheet and in my lab summary. SJK 01:08, 22 December 2010 (EST)

This was a great thing to try! It doesn't prove it, of course, but is evidence that should be pursued if data were to be retaken.