BME100 s2014:T Group10 L3
| Home People Lab Write-Up 1 | Lab Write-Up 2 | Lab Write-Up 3 Lab Write-Up 4 | Lab Write-Up 5 | Lab Write-Up 6 Course Logistics For Instructors Photos Wiki Editing Help | ||||||
OUR TEAM
LAB 3A WRITE-UPDescriptive Statistics
ResultsAll Error Bars are Standard Deviation
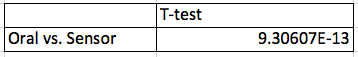
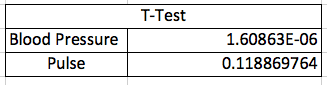
AnalysisA Pearson's R test was conducted between the oral temperature sensor and the RAIIN device and between the blood pressure results of the watch sensor and the blood pressure cuff, and the pulse results from the watch sensor with the Pulse Oximeter device. It was necessary to do a Pearson's R test to determine the correlation between the results provided by the devices we were testing as compared with those of the gold standard devices. For our purposes, a large correlation would indicate that the new device provided very correlated data compared with the gold standard device, and was thusly probably pretty good at measuring that information. As you can see, the RAIIN device offered only a small correlation with the oral thermometer in the laboratory, but no correlation during outside testing. Overall, the device showed no correlation to the oral thermometer. The watch sensor offered a small correlation to the blood pressure cuff in determining blood pressure measurements, but had a large correlation with the Pulse Oximeter in regards to the pulse data. This begins to show the researchers that this device is decent at measuring blood pressure, and pretty good at measuring pulse.
We ran three Student's T-Tests on our data to determine if there were statistically significant differences between the measurements of the RAIIN sensor and oral thermometer, the watch sensor and the blood pressure cuff, and the watch sensor and the Pulse Oximeter. We used T-Tests instead of ANOVA ones because there are only two groups for each test. Also, the T-Test is paired because it was the same student taking the measurements with both devices. A Bonferroni correction was not necessary since we did only a T-Test, and not an ANOVA. The P values of the temperature data and the blood pressure data are below the 0.05 cutoff, leading us to believe that there is less than a 95% chance that we are not wrong in our determination. However, the pulse data's P value is above this threshold, so we are unable to declare the same level of certainty with the pulse data.
Summary/DiscussionThere can be many different flaws with design of any device. In particular, the flaws with a blood pressure wrist cuff and a RAIIN sensor using the Vitals Monitor App installed on an iPhone can be connected to the parts, the energy source, and even the directions to use the device. If any of the parts in or on the device are removable or replaceable they may be positioned incorrectly. The battery can be misplaced or the cuff could be positioned on the person to give faulty readings. A better choice would be for a device to have a more intuitive design that is easy to understand for the user and difficult to get wrong. In addition to faulty readings from positioning, the readings can be off because of the mechanical parts or the connectivity of the program for sensors with the Vitals Monitor Application that is installed on the iPhone. Although BlueTooth is a convenient and popular technology, the unreliability and complicated setup of the RAIIN device makes us think that sacrificing wireless connectivity via BlueTooth for a more reliable system that requires less upkeep. The energy source also is an area for concern. The drain on the battery should not be excessive and the use of the device should have a reasonable amount of usage before they need to be replaced. Using a wired device would alleviate the burden on the battery. The most important plan for the device should be easy usage. If the device itself cannot be explained simply with written directions, the accuracy of the device will never be evident. An improved device should have simple, clear instructions available in the device's packaging as well as online and embedded with any app that uses the device. Another flaw could be in the storage of the data over a period of time. Accuracy matters in all devices that monitor health vitals. Furthermore, questions of privacy and data integrity come to mind whenever something private like healthcare meets something notorious for privacy violations, like technology and the internet. We would need to consider the protection of our user's privacy as one of our chief concerns if we were to design a product that utilizes cloud-based or online storage of personal data. A flaw in design can lead to disaster. All of these things are critical to producing a quality device that can better humanity.
LAB 3B WRITE-UPTarget Population and NeedOur target population is active people that want a better, more intuitive way of measuring their heart rate and blood pressure to track the efficacy of their workout and active lifestyle along with a way of sharing this information with their friends. While there are devices on the market that can measure these vitals already, they are actual physical devices the user must carry around for them to work. Face the Facts by HeartOn Industries uses software developed by MIT measures a person's pulse and respiration rate by exaggerating the minuscule movements and color changes in a person's face. All the user has to do is open the Face the Facts app and take a second of video of your face and their vitals will be shown in real time. Face the Facts, however, piggy-backs off the device you already carry around with you, your phone, so you don't have another device to worry about. Our cutting-edge software can determine a your pulse and respiration rate simply by taking a video of your face. In the future, HeartOn industries can expand our target population to include people with blood circulation problems, because our app can gather localized blood circulation data. Also, future updates may include physician integration for data sharing and patient tracking.
Device DesignFace the Facts by HeartOn Industries uses software developed by MIT measures a person's pulse and respiration rate by exaggerating the minuscule movements and color changes in a person's face. All the user has to do is open the Face the Facts app and take a second of video of their face and their vitals will be shown in real time.
Inferential StatisticsAs you can see from our inferential statistics, our device is very similar to the gold standards in measuring heart rate and respiration. Also, there is a strong positive correlation between our device and the gold standards. In fact, our device is so good, it's almost like we made the data up! Just kidding, that'd be crazy!
Graph
|
||||||