User:Megan L. Channell/Notebook/Horseradish/2013/09/03: Difference between revisions
From OpenWetWare
No edit summary |
No edit summary |
||
| Line 29: | Line 29: | ||
#For each sample, 1mL of the sample was pipetted into a clean cuvette | #For each sample, 1mL of the sample was pipetted into a clean cuvette | ||
# The sample was returned to the vial it was kept in and the cuvette was rinsed three times with DI water | # The sample was returned to the vial it was kept in and the cuvette was rinsed three times with DI water | ||
[[Image:Cmj conc 9 3.jpg|1050px]] | |||
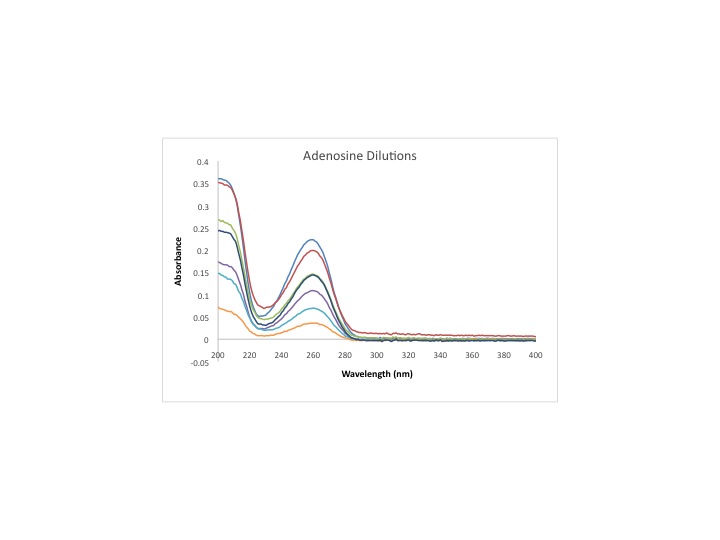
==Data== | ==Data== | ||
A UV-Vis was ran from 800-200nm. The absorbance and calibration curve were done through the data collected on the UV-Vis. | A UV-Vis was ran from 800-200nm. The absorbance and calibration curve were done through the data collected on the UV-Vis. | ||
[[Image:Adenosine1 9 3 2013 cmj.jpg|1050px]] | |||
<!-- ##### DO NOT edit below this line unless you know what you are doing. ##### --> | <!-- ##### DO NOT edit below this line unless you know what you are doing. ##### --> | ||
Revision as of 13:48, 8 September 2013
 Biomaterials Design Lab Biomaterials Design Lab
|
<html><img src="/images/9/94/Report.png" border="0" /></html> Main project page <html><img src="/images/c/c3/Resultset_previous.png" border="0" /></html>Previous entry<html> </html>Next entry<html><img src="/images/5/5c/Resultset_next.png" border="0" /></html> |
Adenosine and Inosine UV-VisObjectiveFinding the molar absorptivities of adenosine and inosine. This will help later on in studying adenosine deaminase (ADA) which converts adenosine to inosine.. The structural differences between the two molecules is that adenosine has a primary amine while inosine has a carboxy group. ProtocolStock Solutions Two different stocks solutions were made, one adenosine and one inosine
Dilutions A total of 14 dilutions were made
A sample calculation: Amount of Stock Solution.= [10 mL(dilution concentration)/(stock solution concentration)] UV-Vis
DataA UV-Vis was ran from 800-200nm. The absorbance and calibration curve were done through the data collected on the UV-Vis.
| |