Kafatos:Research: Difference between revisions
No edit summary |
|||
| (6 intermediate revisions by 2 users not shown) | |||
| Line 1: | Line 1: | ||
{{Kafatos/Christophides Lab}} | {{Kafatos/Christophides Lab}} | ||
__NOTOC__ | |||
{|cellspacing="5" cellpadding="10" style="background:#00538D; width: 800px;" | |||
|-valign="top" | |||
|style="background:#ffffff"| | |||
==Functional genomics of mosquito vector/malaria parasite interactions== | ==Functional genomics of mosquito vector/malaria parasite interactions== | ||
<blockquote> | |||
The [[Kafatos:Kafatos/Christophides Lab|Laboratory of Insect Immunogenomics]] focuses on the study of the major vector of malaria in Africa, ''Anopheles gambiae'' and particularly how its innate immune system manipulates and is manipulated by the malaria parasite during its passage through the mosquito. Malaria is one of the main infectious causes of human mortality world-wide, mainly among young children in sub-Saharan Africa. Transmission of the malaria agent, the ''Plasmodium'' parasite, requires its cyclical development in two organisms: the human host and the ''Anopheles'' mosquito vector. Current disease control methods that aim to either cure the disease in the human body or to control the vector populations are hampered due to increase in drug resistance of the parasite and insecticide resistance of the mosquito. Our underlying conviction is that bringing the power of functional genomics into the study of this biological system will rapidly advance our understanding of mosquito immunity and parasite development. This will be crucial for the development of novel approaches to malaria control that are urgently needed to reinforce and complement the ongoing research into drug, vaccine and insecticide development.<BR> | |||
Completion of the genome sequence of ''A. gambiae'' two years ago, together with the development of DNA microarrays in this species and adaptation of the RNAi technique to adult mosquitoes, has allowed comparative and functional genomic approaches to understanding the mosquito innate immune system, and its interactions with parasites. Using the rodent model system, ''P. berghei'', we have identified a variety of factors that negatively affect the development of parasites in the mosquito (antagonists), in some cases leading to complete transmission blockage. In addition, mosquito molecules have been identified that play positive roles and are required for successful parasite transmission (agonists). Research is continuing to identify new factors involved in these interactions and to decipher the interplay of these molecules and their regulation. Importantly, as our findings indicate a highly complex interplay between parasite and vector, we are currently extending our studies of parasite-vector interactions towards the human malaria parasite, ''P. falciparum''. | |||
</blockquote> | |||
|} | |||
{|cellspacing="5" cellpadding="10" style="background:#00538D; width: 800px;" | |||
|-valign="top" | |||
|style="background:#ffffff"| | |||
==Genomic approaches== | ==Genomic approaches== | ||
<blockquote> | |||
To investigate the genetic armory of the ''A. gambiae'' mosquito we have initially used an EST library derived from two immune competent mosquito cell lines to construct DNA microarrays containing approximately 2,500 genes <cite>dimopoulos2002</cite>. These arrays permitted for the first time an understanding of the global innate immune responses in adult mosquitoes and cultured cells, as well as the mosquito reaction to ''Plasmodium'' infection and mechanisms for refractoriness to the parasite. To increase further the resolution of this powerful technique, we recently constructed DNA microarrays containing approximately 20,000 ''Anopheles'' ESTs representing over 9,000 mosquito genes, and work is ongoing to develop an amplicon-based microarray platform encompassing all predicted genes in the mosquito genome. This resource also allows for high throughput production of dsRNAs that can be used for RNAi gene silencing as well as expression of fragments of all predicted mosquito proteins, for use in antibody production.<BR> | To investigate the genetic armory of the ''A. gambiae'' mosquito we have initially used an EST library derived from two immune competent mosquito cell lines to construct DNA microarrays containing approximately 2,500 genes <cite>dimopoulos2002</cite>. These arrays permitted for the first time an understanding of the global innate immune responses in adult mosquitoes and cultured cells, as well as the mosquito reaction to ''Plasmodium'' infection and mechanisms for refractoriness to the parasite. To increase further the resolution of this powerful technique, we recently constructed DNA microarrays containing approximately 20,000 ''Anopheles'' ESTs representing over 9,000 mosquito genes, and work is ongoing to develop an amplicon-based microarray platform encompassing all predicted genes in the mosquito genome. This resource also allows for high throughput production of dsRNAs that can be used for RNAi gene silencing as well as expression of fragments of all predicted mosquito proteins, for use in antibody production.<BR> | ||
| Line 27: | Line 36: | ||
#vlachou2004 pmid=15186403 Real-time, in vivo analysis of malaria ookinete locomotion and mosquito midgut invasion. | #vlachou2004 pmid=15186403 Real-time, in vivo analysis of malaria ookinete locomotion and mosquito midgut invasion. | ||
</biblio> | </biblio> | ||
< | </blockquote> | ||
|} | |||
{|cellspacing="5" cellpadding="10" style="background:#00538D; width: 800px;" | |||
|-valign="top" | |||
|style="background:#ffffff"| | |||
==Targeted approaches== | ==Targeted approaches== | ||
<blockquote> | |||
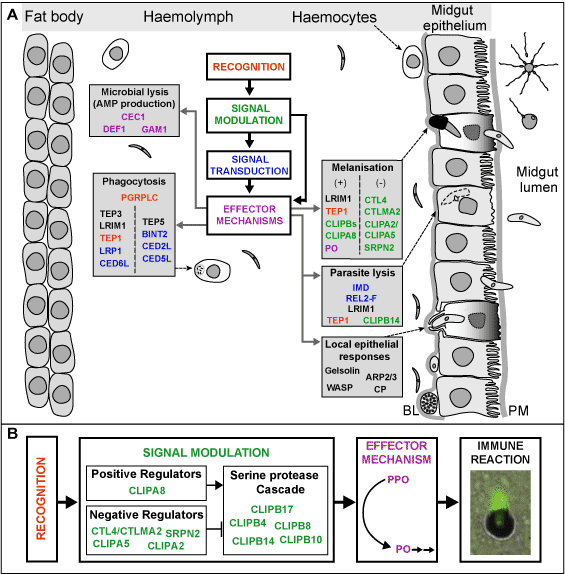
Dissecting the mosquito immune pathways. Pioneering studies in the fly ''Drosophila melanogaster'' contributed to the detailed understanding of innate immunity and showed that the underlying mechanisms have largely been conserved through the course of metazoan evolution <cite>hoffmann2002</cite>. These studies revealed most of the components of two conserved immune signaling pathways, ''Toll'' and ''Imd'', that are utilized by the fly to respond to bacterial and fungal infections (Figure 2A). The availability of the ''A. gambiae'' genome sequence <cite>holt2002</cite> previously allowed us to perform a comparative genomic analysis of putative immunity genes between ''Anopheles'' and ''Drosophila'' <cite>christophides2002</cite>. Although the majority of the intracellular components of the ''Toll'' and ''Imd'' pathways are conserved between the two organisms (Figure 2B), a number of differences – which may have significant impact on immunity mechanisms – were also identified. The most important of these is the absence of the NF-kB-like transcription factor ''Dif'' in ''Anopheles''. This suggested that in a functional ''Toll'' pathway either ''REL1'' (the ortholog of ''Dorsal'') or ''REL2'' (the ortholog of ''Relish'') might substitute for ''Dif''. We investigated the role of REL2 and other molecules that are possibly implicated in the same signalling pathway in the mosquito immune responses. REL2 regulates the inducible expression of the various antimicrobial peptide genes including ''CEC1'' and the key parasite antagonist, ''LRIM1'' <cite>osta2004</cite>. We showed that the ''REL2'' gene is alternatively spliced, resulting in two protein isoforms that are differentially implicated in defense against Gram-positive or Gram-negative bacteria. Thus, through alternative splicing ''Anopheles'' uses a single gene to mediate reactions for which ''Drosophila'' employs two genes, ''Relish'' and ''Dif''. The ''REL2'' pathway is also involved in the control of ''Plasmodium'' parasite infection of the mosquito midgut. Silencing of the pathway drastically increases the parasite numbers that successfully develop into oocysts.<BR> | Dissecting the mosquito immune pathways. Pioneering studies in the fly ''Drosophila melanogaster'' contributed to the detailed understanding of innate immunity and showed that the underlying mechanisms have largely been conserved through the course of metazoan evolution <cite>hoffmann2002</cite>. These studies revealed most of the components of two conserved immune signaling pathways, ''Toll'' and ''Imd'', that are utilized by the fly to respond to bacterial and fungal infections (Figure 2A). The availability of the ''A. gambiae'' genome sequence <cite>holt2002</cite> previously allowed us to perform a comparative genomic analysis of putative immunity genes between ''Anopheles'' and ''Drosophila'' <cite>christophides2002</cite>. Although the majority of the intracellular components of the ''Toll'' and ''Imd'' pathways are conserved between the two organisms (Figure 2B), a number of differences – which may have significant impact on immunity mechanisms – were also identified. The most important of these is the absence of the NF-kB-like transcription factor ''Dif'' in ''Anopheles''. This suggested that in a functional ''Toll'' pathway either ''REL1'' (the ortholog of ''Dorsal'') or ''REL2'' (the ortholog of ''Relish'') might substitute for ''Dif''. We investigated the role of REL2 and other molecules that are possibly implicated in the same signalling pathway in the mosquito immune responses. REL2 regulates the inducible expression of the various antimicrobial peptide genes including ''CEC1'' and the key parasite antagonist, ''LRIM1'' <cite>osta2004</cite>. We showed that the ''REL2'' gene is alternatively spliced, resulting in two protein isoforms that are differentially implicated in defense against Gram-positive or Gram-negative bacteria. Thus, through alternative splicing ''Anopheles'' uses a single gene to mediate reactions for which ''Drosophila'' employs two genes, ''Relish'' and ''Dif''. The ''REL2'' pathway is also involved in the control of ''Plasmodium'' parasite infection of the mosquito midgut. Silencing of the pathway drastically increases the parasite numbers that successfully develop into oocysts.<BR> | ||
| Line 60: | Line 75: | ||
#ligoxygakis2002 pmid=12456640 | #ligoxygakis2002 pmid=12456640 | ||
</biblio> | </biblio> | ||
<BR><BR> | </blockquote> | ||
|} | |||
{|cellspacing="5" cellpadding="10" style="background:#00538D; width: 800px;" | |||
|-valign="top" | |||
|style="background:#ffffff"| | |||
==BBSRC Research Grant on:<BR> Genomic analysis of NF-kappaB signalling in Anopheles gambiae== | |||
<blockquote> | |||
The innate immune system is the first line of defence against infections in higher organisms. In insects, which like other invertebrate animals lack adaptive immune systems, innate immunity is the only defence system. Innate immunity relies on receptors that recognize specific molecular structures shared between microbes or danger signals generated during an infection. Recognition triggers specific signalling pathways, cellular processes or enzymatic cascades, which activate various effector mechanisms that combat infections. In many cases, the effector mechanisms require de novo production of effector or regulatory proteins, which is usually controlled by transcription factors of the Rel/Nuclear Factor/kappaB (NF-kappaB) family. This BBSRC-funded project aims to dissect the mechanisms of gene expression that are under the control of NF-kappaB signalling pathways in the African mosquito Anopheles gambiae. This mosquito is a vector for animal and human diseases including malaria that is caused by the protozoan parasite Plasmodium. In recent years, thanks to the availability of its genome sequence and development of robust genetics and genomics tools to investigate the gene function, A. gambiae has become a model system to study the interactions between microbes, especially Plasmodium, and the innate immune system.<BR><BR> | |||
We had previously shown that the mosquito REL2 signalling pathway, which is equivalent to the Drosophila Imd, is responsible for limiting infections of the rodent malaria parasite Plasmodium berghei and that the same pathway is required for resistance against infections with gram-positive and gram-negative bacteria (Meister et al., PNAS, 2005). This project significantly advanced our understanding of the molecular mechanisms that regulate these reactions. We have shown that these two phenomena are tightly linked (Meister et al., PLoS Pathogens, 2009). Infections with bacteria are sensed by the peptidoglycan recognition receptor PGRPLC and activate the REL2 pathway. In this way, the REL2 pathway controls the size of bacteria populations residing in the mosquito midgut, which dramatically increase soon after ingestion of blood. In addition to limiting the bacterial infection, downstream effectors of this pathway also act upon Plasmodium ookinetes invading the mosquito gut, killing a substantial fraction of them. Importantly, this mechanism reduces mosquito infections with field isolates of the deadliest of the human malaria parasites, Plasmodium falciparum, as well as with the laboratory model parasite, Plasmodium berghei. These findings open new research avenues towards understanding the mosquito/parasite interactions, in which symbiotic microbes play a central modulatory role.<BR><BR> | |||
Two of the downstream effectors of the mosquito NF-kappaB pathways are the leucine-rich repeat proteins LRIM1 and APL1C. We and others had previously shown that these hemolymph proteins are important antagonists of Plasmodium infections. In the context of this project we have generated substantial new knowledge of the molecular mechanisms implicated in this process (Povelones et al., Science, 2009). We have shown that LRIM1 and APL1C circulate in the mosquito hemolymph as a complex that physically interacts with the complement C3-like protein, TEP1, promoting its cleavage and/or stabilization and localization on the surface of parasites, ultimately causing their lysis or melanization. These data establish a novel mechanism of complement pathway activation in insects. | |||
To identify novel regulators of the two NF-kappaB pathways, we have establish high throughput cell-based RNAi screens of over 100 genes expressed in mosquito haemocytes or transcriptionally induced following bacterial infections (Lombardo et al., MPM Proceedings, 2009). We have cloned, tested and used the promoter of the LRIM1 gene, as well as the promoter of the antimicrobial peptide encoding gene CEC1, in luciferase based reporter assays following challenge with various elicitors. Our data have identified several novel regulators of these pathways that are currently being investigated.<BR><BR> | |||
Finally, in collaboration with the Broad Institute and the Harvard School of Public Health we have developed a novel high-throughput genome-wide genotyping tool (Muskavitch et al., ASTMH Proceedings, 2008). Development of this SNP chip has been enabled by the discovery of abundant single nucleotide polymorphisms (SNPs) through resequencing of multiple strains by a multicentre consortium. The array is based on a filtered set of 400000 SNP assays, 66000 of which yield perfectly accurate SNP calls. Pooled hybridization results using material from multiple mosquitoes exhibit a very high correlation with averaged hybridization results from individual mosquitoes, enabling the use of a single chip to quantitatively describe genome-wide allele frequencies in a large sample of mosquitoes. We expect that this tool will be widely adopted among vector biologists with interests in mapping of genes underlying important traits in A. gambiae and in analysis of mosquito population structure. | |||
</blockquote> | |||
|} | |||
{|cellspacing="5" cellpadding="10" style="background:#00538D; width: 800px;" | |||
|-valign="top" | |||
|style="background:#ffffff"| | |||
==Malaria and mosquito population dynamics== | ==Malaria and mosquito population dynamics== | ||
<blockquote> | |||
Transmission of the ''Plasmodium'' parasite to a human host completely depends on the availability of a competent mosquito vector. ''A. gambiae'' is the most important and efficient vector for transmission of human malaria in Africa. In some cases, ''A. gambiae'' kills the ''Plasmodium'' parasite, thus blocking the transmission cycle. The interactions between the vector and the parasite involve the mosquito’s innate immunity. Sequence polymorphisms occurring in immune-related genes (as already documented for ''TEP1'') may reflect phenotypic variations in vector competence. Moreover polymorphism may be indicative of adaptation in a co-evolving mosquito/parasite system. We therefore screened four strains of ''A. gambiae'' (susceptible and refractory to ''Plasmodium'' parasites) and wild populations for their polymorphism. Sixty immune-related genes were sequenced, and Single Nucleotide Polymorphisms (SNPs) were identified by alignment. The diversity in immune-related genes was high compared to other parts of the genome, suggesting a diversifying selection acting on these sequences. The identified SNPs will be used to investigate the potential association between genotypes and the susceptibility/refractoriness of ''A. gambiae'' to the parasite under field conditions. | Transmission of the ''Plasmodium'' parasite to a human host completely depends on the availability of a competent mosquito vector. ''A. gambiae'' is the most important and efficient vector for transmission of human malaria in Africa. In some cases, ''A. gambiae'' kills the ''Plasmodium'' parasite, thus blocking the transmission cycle. The interactions between the vector and the parasite involve the mosquito’s innate immunity. Sequence polymorphisms occurring in immune-related genes (as already documented for ''TEP1'') may reflect phenotypic variations in vector competence. Moreover polymorphism may be indicative of adaptation in a co-evolving mosquito/parasite system. We therefore screened four strains of ''A. gambiae'' (susceptible and refractory to ''Plasmodium'' parasites) and wild populations for their polymorphism. Sixty immune-related genes were sequenced, and Single Nucleotide Polymorphisms (SNPs) were identified by alignment. The diversity in immune-related genes was high compared to other parts of the genome, suggesting a diversifying selection acting on these sequences. The identified SNPs will be used to investigate the potential association between genotypes and the susceptibility/refractoriness of ''A. gambiae'' to the parasite under field conditions. | ||
< | </blockquote> | ||
|} | |||
Latest revision as of 06:53, 25 May 2010
| Click here to visit our NEW WEBSITE |
| The content below is most likely out of date. We also have a new lean and mean openwetware area. |
Functional genomics of mosquito vector/malaria parasite interactions
|
Genomic approaches
|
Targeted approaches
|
BBSRC Research Grant on:
|
Malaria and mosquito population dynamics
|