Haynes Lab:Notebook/Characterizing AHL quorum sensing homologs/2013/10/28: Difference between revisions
From OpenWetWare
Jump to navigationJump to search
Rene M Davis (talk | contribs) |
Rene M Davis (talk | contribs) |
||
| Line 21: | Line 21: | ||
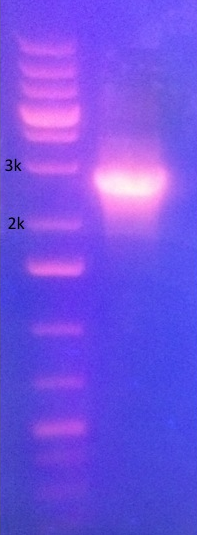
PCR ''need to edit this to include the next lane. lane 1 is P087/P088 and lane 2 is P089/P090, both I13522'' [[image:13.10.28_PCR_cropped.png|PCR]] | PCR ''need to edit this to include the next lane. lane 1 is P087/P088 and lane 2 is P089/P090, both I13522'' [[image:13.10.28_PCR_cropped.png|PCR]] | ||
<br><br> | <br><br> | ||
Ran another PCR reaction to text which primer wasn't working for the sender backbone. Used the two sender backbone primers, P089 and P090 with the matching F or R primer from the pLux vectors primers which were shown to work. Rxn 3 is positive control. | Ran another PCR reaction to text which primer wasn't working for the sender backbone. Used the two sender backbone primers, P089 and P090 with the matching F or R primer from the pLux vectors primers which were shown to work. Rxn 3 is positive control.<br> | ||
1 | 1 | ||
:15ul reaction | :15ul reaction | ||
Revision as of 15:23, 15 November 2013
| <html><img src="/images/9/94/Report.png" border="0" /></html> Main project page <html><img src="/images/c/c3/Resultset_previous.png" border="0" /></html>Previous entry<html> </html>Next entry<html><img src="/images/5/5c/Resultset_next.png" border="0" /></html> | |
mm/dd/yyyyMiniprepped J04450 that I started growing on Saturday. They were in the incubator for a long time and were super red.
Gel of cut J04450 PCR need to edit this to include the next lane. lane 1 is P087/P088 and lane 2 is P089/P090, both I13522
2
3
| |