Frankel:Force Spectroscopy: Difference between revisions
No edit summary |
No edit summary |
||
| Line 20: | Line 20: | ||
| align="justify" rowspan="2" width="70%" style="border: 2px solid #000033; background-color:#ffffff; padding:1em;" valign="top"| | | align="justify" rowspan="2" width="70%" style="border: 2px solid #000033; background-color:#ffffff; padding:1em;" valign="top"| | ||
'''<font color=#000000 font size=3>Self assembly and pore formation of HIV GP160 revealed at molecular resolution</font>''' | |||
[[Image:Stlow.png|300px]] | [[Image:Stlow.png|300px]] | ||
Revision as of 19:45, 8 November 2012
<owwmenu align="center" font="helvetica" bold="1" color="white" bgcolor="black" hovercolor="black" bghovercolor="orange" topfontsize="10" fontSize="10" image="Danbanner-bio-machines.jpg" >
Home=Frankel Members=#,Principal Investigator=Frankel:Lab_Members, PhD students=Frankel:Lab_Members, Alumni=Frankel:Lab_Members Contact=Frankel:Contact Collaborators=Frankel:Collaborators Publications=Frankel:Publications Lab=Frankel:Research Research=#,Force Spectroscopy=Frankel:Force Spectroscopy,HIV/Virus=Frankel:HIV/Virus,ECM Proteins=Frankel:ECM Proteins,Cyberplasm=Frankel:Cyberplasm,Cancer=Frankel:Cancer
<startFeed/>
Force Spectroscopy
|
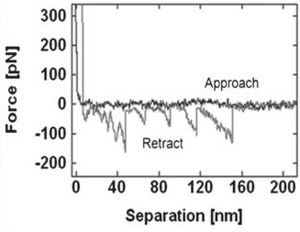
Force spectra taken on raised terraces and lower features. Rupture force distribution of self assembled gp160 unfolding on terraced and lower regions. Rupture forces were measured as 79.6 ± 3.9 pN and 81.3 ± 3.8 pN for the terraces and lower regions, respectively. These forces are much lower than those measured for unfolding of isolated proteins on mica, which were above 160 pN. The lower unfolding forces suggest that GP160 is considerably easier to unfold when aggregated than isolated.
| ||
|
Sawtooth pattern on the retraction force curve indicating the unfolding of fibronectin. The average rupture force distribution of the protein on mica surface was 85.1 ± 2.7 pN. |