Biomod/2013/Aarhus/Supplementary/Supplementary Data: Difference between revisions
No edit summary |
|||
| (45 intermediate revisions by 4 users not shown) | |||
| Line 5: | Line 5: | ||
===Origami design=== | ===Origami design=== | ||
[[image:origami 3.png|center|frame|Details of the different modules of the plate.]] | [[image:origami 3.png|center|frame|Table S1. Details of the different modules of the plate.]] | ||
<html><br style="clear:both;"/></html> | |||
[[image:origami 8.png|center|frame|Details of the different modules.]] | [[Biomod/2013/Aarhus/Supplementary/Supplementary_Informations#Staple_strands_for_the_plate|Staple strand sequences for the plate]] see supplementary information. | ||
[[image:origami 7.png|center|frame|Table S2. Details of the dome structure.]] | |||
<html><br style="clear:both;"/></html> | |||
[[image:origami 8.png|center|frame|Table S3.Details of the different dome modules.]] | |||
<html><br style="clear:both;"/></html> | |||
[[Biomod/2013/Aarhus/Supplementary/Supplementary_Informations#Staple_strands_for_the_dome|Figure and staple strand sequences for the dome]] see supplementary information. | |||
===sisiRNA=== | ===sisiRNA=== | ||
| Line 15: | Line 24: | ||
====Yields of RP-HPLC purifications of the modified strands==== | ====Yields of RP-HPLC purifications of the modified strands==== | ||
[[image:table sisi 1.png|center|frame|Table | [[image:table sisi 1.png|center|frame|Table S4: Yields of RP-HPLC purifications of the 12 modified strands.]] | ||
<html><br style="clear:both;"/></html> | |||
====Estimated yields of the annealing reactions==== | ====Estimated yields of the annealing reactions==== | ||
[[image:table sisi 2.png|center|frame|Estimated yields of the annealing reactions.]] | [[image:table sisi 2.png|center|frame|Table S5. Estimated yields of the annealing reactions.]] | ||
<html><br style="clear:both;"/></html> | |||
====Luciferase knockdown with all singly modified duplexes==== | ====Luciferase knockdown with all singly modified duplexes==== | ||
[[image:sisi sup 3.png| | [[image:sisi sup 3.png|thumb|900px|Figure S1. Cell viability corrected luciferase activities for all singly modified duplexes, transfected with Lipofectamine. Click on the image and choose 'high resolution' for details.]] | ||
[[image:sisi supp 5.png| | [[image:sisi supp 4.png|center|frame|Table S6. Best fit IC50 values of all knockdown experiments using singly modified duplexes using Lipofectamine.The IC50 value for W179-PEG1K was calculated with a low R2 value and is not conclusive.]] | ||
<html><br style="clear:both;"/></html> | |||
[[image:sisi supp 5.png|thumb|900px|Figure S2. Luciferase and MTT measurements of all knockdown experiments with singly modified duplexes using Lipofectamine.]] | |||
===System in action=== | ===System in action=== | ||
====Radioactive labeling of W376==== | ====Radioactive labeling of W376==== | ||
[[image:sys sisi 1.png|center|frame| | [[image:sys sisi 1.png|center|frame|Figure S3. 1) 25 bp DNA ladder, 2) 6 µL labeled W376, 3) 1 pmol W376, 4) 1.5 pmol W376, 5) pmol W376, 6) 2.5 pmol W376, 7) pmol W376.]] | ||
<html><br style="clear:both;"/></html> | |||
The band containing the labeled sample appears most similar to that in lane 4 which means that the 6 µL that were run on the gel contained 1.5 pmol W376, corresponding to a concentration of 0.25 µM in the purified sample. | The band containing the labeled sample appears most similar to that in lane 4 which means that the 6 µL that were run on the gel contained 1.5 pmol W376, corresponding to a concentration of 0.25 µM in the purified sample. | ||
===Cholesterol=== | ===Cholesterol=== | ||
'''<sup>1</sup>H NMR, <sup>13</sup>C NMR and IR of compound 4.''' | |||
'''<sup>1</sup>H NMR | <html><A name="NMR4H"/></html> | ||
[[Image:Biomod_Aarhus_Chem_NMR_4H.png|900x900px|Center|thumb|<sup>1</sup>H NMR 0f compound 4]] | |||
<html><br style="clear:both;"/></html> | |||
<html><A name="NMR4C"/></html> | |||
[[Image:Biomod_Aarhus_Chem_NMR_4C1.png|900x900px|Center|thumb|<sup>13</sup>C NMR of compound 4]] | |||
<html><br style="clear:both;"/></html> | |||
[[Image:Biomod_Aarhus_Chem_NMR_4C2.png|900x900px|Center|thumb|Zoom of <sup>13</sup>C NMR of compound 4]] | |||
<html><br style="clear:both;"/></html> | |||
<html><A name="IR4"/></html> | |||
[[Image:Biomod_Aarhus_Chem_IR4.png|900x900px|Center|thumb|IR of compound 4]] | |||
<html><br style="clear:both;"/></html> | |||
'''<sup>1</sup>H NMR, <sup>13</sup>C NMR and IR of compound 5.''' | |||
[[Image: | <html><A name="NMR5H"/></html> | ||
[[Image: | [[Image:Biomod_Aarhus_Chem_NMR_5H.png|900x900px|Center|thumb|<sup>1</sup>H NMR of compound 5]] | ||
[[Image: | <html><br style="clear:both;"/></html> | ||
<html><A name="NMR5C"/></html> | |||
[[Image:Biomod_Aarhus_Chem_NMR_5C1.png|900x900px|Center|thumb|<sup>13</sup>C NMR of compound 5]] | |||
<html><br style="clear:both;"/></html> | |||
[[Image:Biomod_Aarhus_Chem_NMR_5C2.png|900x900px|Center|thumb|Zoom of <sup>13</sup>C NMR of compound 5]] | |||
<html><br style="clear:both;"/></html> | |||
<html><A name="IR5"/></html> | |||
[[Image:Biomod_Aarhus_Chem_IR5.png|900x900px|Center|thumb|IR of compound 5]] | |||
<html><br style="clear:both;"/></html> | |||
'''<sup>1</sup>H NMR and <sup>13</sup>C NMR of compound 6.''' | |||
<html><A name="NMR6H"/></html> | |||
[[Image:Biomod_Aarhus_Chem_NMR_6H.png|900x900px|Center|thumb|<sup>1</sup>H NMR of compound 6]] | |||
<html><br style="clear:both;"/></html> | |||
<html><A name="NMR6C"/></html> | |||
[[Image:Biomod_Aarhus_Chem_NMR_6C.png|900x900px|Center|thumb|<sup>13</sup>C NMR of compound 6]] | |||
<html><br style="clear:both;"/></html> | |||
[[Image:Biomod_Aarhus_Chem_NMR_6C2.png|900x900px|Center|thumb|Zoom of <sup>13</sup>C NMR of compound 6]] | |||
<html><br style="clear:both;"/></html> | |||
===Photosensitizer=== | ===Photosensitizer=== | ||
'''<sup>1</sup>H NMR and <sup>13</sup>C NMR of compound 7.''' | |||
<html><A name="NMR7H"/></html> [[Image:Biomod_Aarhus_Chem_NMR_7H.png|900x900px|Center|thumb|<sup>1</sup>H NMR of compound '''7''']] | |||
<html><br style="clear:both;"/></html> | |||
<html><A name="NMR7C"/></html> [[Image:Biomod_Aarhus_Chem_NMR_7C.png|900x900px|Center|thumb|<sup>13</sup>C NMR of compound '''7''']] | |||
<html><br style="clear:both;"/></html> | |||
'''<sup>1</sup>H NMR and <sup>13</sup>C NMR of compound 8.''' | |||
<html><A name="NMR8H"/></html> [[Image:Biomod_Aarhus_Chem_NMR_8H.png|900x900px|Center|thumb|<sup>1</sup>H NMR of compound '''8''']] | |||
<html><br style="clear:both;"/></html> | |||
<html><A name="NMR8C"/></html> [[Image:Biomod_Aarhus_Chem_NMR_8C.png|900x900px|Center|thumb|<sup>13</sup>C NMR of compound '''8''']] | |||
<html><br style="clear:both;"/></html> | |||
'''<sup>1</sup>H NMR, <sup>13</sup>C NMR and UV of compound 9.''' | |||
<html><A name="NMR9H"/></html> [[Image:Biomod_Aarhus_Chem_NMR_9H.png|900x900px|Center|thumb|<sup>1</sup>H NMR of compound '''9''']] | |||
<html><br style="clear:both;"/></html> | |||
<html><A name="NMR9C"/></html> [[Image:Biomod_Aarhus_Chem_NMR_9C.png|900x900px|Center|thumb|<sup>13</sup>C NMR of compound '''9''']] | |||
<html><br style="clear:both;"/></html> | |||
<html><A name="9UV"/></html> [[Image:Biomod_Aarhus_Chem_10UV.png|900x900px|Center|UV of compound '''9''']] | |||
<html><br style="clear:both;"/></html> | |||
'''<sup>1</sup>H NMR | '''<sup>1</sup>H NMR, <sup>13</sup>C NMR and UV of compound 10.''' | ||
[[Image:Biomod_Aarhus_Chem_NMR_10H.png| | <html><A name="NMR10H"/></html> [[Image:Biomod_Aarhus_Chem_NMR_10H.png|900x900px|Center|thumb|<sup>1</sup>H NMR of compound '''10''']] | ||
[[Image:Biomod_Aarhus_Chem_NMR_10C.png| | <html><br style="clear:both;"/></html> | ||
<html><A name="NMR10C"/></html> [[Image:Biomod_Aarhus_Chem_NMR_10C.png|900x900px|Center|thumb|<sup>13</sup>C NMR of compound '''10''']] | |||
<html><br style="clear:both;"/></html> | |||
<html><A name="10UV"/></html> [[Image:Biomod_Aarhus_Chem_11UV.png|900x900px|Center|UV of compound '''10''']] | |||
<html><br style="clear:both;"/></html> | |||
'''<sup>1</sup>H NMR and <sup>13</sup>C NMR | '''<sup>1</sup>H NMR and <sup>13</sup>C NMR of compound 11.''' | ||
[[Image:Biomod_Aarhus_Chem_NMR_11H.png| | <html><A name="NMR11H"/></html> [[Image:Biomod_Aarhus_Chem_NMR_11H.png|900x900px|Center|thumb|<sup>1</sup>H NMR of compound '''11''']] | ||
[[Image:Biomod_Aarhus_Chem_NMR_11C.png| | <html><br style="clear:both;"/></html> | ||
<html><A name="NMR11C"/></html> [[Image:Biomod_Aarhus_Chem_NMR_11C.png|900x900px|Center|thumb|<sup>13</sup>C NMR of compound '''11''']] | |||
<html><br style="clear:both;"/></html> | |||
'''<sup>1</sup>H NMR and <sup>13</sup>C NMR | '''<sup>1</sup>H NMR and <sup>13</sup>C NMR of compound 12.''' | ||
[[Image:Biomod_Aarhus_Chem_NMR_12H.png| | <html><A name="NMR12H"/></html> [[Image:Biomod_Aarhus_Chem_NMR_12H.png|900x900px|Center|thumb|<sup>1</sup>H NMR of compound '''12''']] | ||
[[Image:Biomod_Aarhus_Chem_NMR_12C.png| | <html><br style="clear:both;"/></html> | ||
<html><A name="NMR12C"/></html> [[Image:Biomod_Aarhus_Chem_NMR_12C.png|900x900px|Center|thumb|<sup>13</sup>C NMR of compound '''12''']] | |||
<html><br style="clear:both;"/></html> | |||
===5-propargylamino-ddUTP=== | ===5-propargylamino-ddUTP=== | ||
'''<sup>1</sup>H NMR, <sup>13</sup>C NMR and IR of compound 15.''' | |||
<html><A name="NMR15H"/></html>[[Image:Biomod_Aarhus_Chem_NMR_15H.png|900x900px|Center|thumb|<sup>1</sup>H NMR of compound 15]] | |||
<html><br style="clear:both;"/></html> | |||
[[Image:Biomod_Aarhus_Chem_NMR_15H2.png|900x900px|Center|thumb|zoom of <sup>1</sup>H NMR of compound 15]] | |||
<html><br style="clear:both;"/></html> | |||
<html><A name="NMR15C"/></html>[[Image:Biomod_Aarhus_Chem_NMR_15C.png|900x900px|Center|thumb|<sup>13</sup>C NMR of compound 15]] | |||
<html><br style="clear:both;"/></html> | |||
<html><A name="IR15"/></html>[[Image:Biomod_Aarhus_Chem_IR15.png|900x900px|Center|thumb|IR of compound 15]] | |||
<html><br style="clear:both;"/></html> | |||
'''<sup>1</sup>H NMR, <sup>13</sup>C NMR and IR of compound 16.''' | |||
<html><A name="NMR16H"/></html>[[Image:Biomod_Aarhus_Chem_NMR_16H.png|900x900px|Center|thumb|<sup>1</sup>H NMR of compound 16]] | |||
<html><br style="clear:both;"/></html> | |||
[[Image:Biomod_Aarhus_Chem_NMR_16H2.png|900x900px|Center|thumb|zoom of <sup>1</sup>H NMR of compound 16]] | |||
<html><br style="clear:both;"/></html> | |||
<html><A name="NMR16C"/></html>[[Image:Biomod_Aarhus_Chem_NMR_16C.png|900x900px|Center|thumb|<sup>13</sup>C NMR of compound 16]] | |||
<html><br style="clear:both;"/></html> | |||
<html><A name="IR16"/></html>[[Image:Biomod_Aarhus_Chem_IR16.png|900x900px|Center|thumb|IR of compound 16]] | |||
<html><br style="clear:both;"/></html> | |||
'''<sup>1</sup>H NMR, <sup>13</sup>C NMR and IR of compound 17.''' | |||
<html><A name="NMR17H"/></html>[[Image:Biomod_Aarhus_Chem_NMR_17H.png|900x900px|Center|thumb|<sup>1</sup>H NMR of compound 17]] | |||
<html><br style="clear:both;"/></html> | |||
[[Image:Biomod_Aarhus_Chem_NMR_17H2.png|900x900px|Center|thumb|zoom of <sup>1</sup>H NMR of compound 17]] | |||
<html><br style="clear:both;"/></html> | |||
<html><A name="NMR17C"/></html>[[Image:Biomod_Aarhus_Chem_NMR_17C.png|900x900px|Center|thumb|<sup>13</sup>C NMR of compound 17]] | |||
<html><br style="clear:both;"/></html> | |||
<html><A name="IR17"/></html>[[Image:Biomod_Aarhus_Chem_IR17.png|900x900px|Center|thumb|IR of compound 17]] | |||
<html><br style="clear:both;"/></html> | |||
'''<sup>1</sup>H NMR, <sup>13</sup>C NMR and IR of compound 18.''' | |||
<html><A name="NMR18H"/></html>[[Image:Biomod_Aarhus_Chem_NMR_18H.png|900x900px|Center|thumb|<sup>1</sup>H NMR of compound 18]] | |||
<html><br style="clear:both;"/></html> | |||
[[Image:Biomod_Aarhus_Chem_NMR_18H2.png|900x900px|Center|thumb|zoom of <sup>1</sup>H NMR of compound 18]] | |||
<html><br style="clear:both;"/></html> | |||
<html><A name="NMR18C"/></html>[[Image:Biomod_Aarhus_Chem_NMR_18C.png|900x900px|Center|thumb|<sup>13</sup>C NMR of compound 18]] | |||
<html><br style="clear:both;"/></html> | |||
<html><A name="IR18"/></html>[[Image:Biomod_Aarhus_Chem_IR18.png|900x900px|Center|thumb|IR of compound 18]] | |||
<html><br style="clear:both;"/></html> | |||
'''<sup>1</sup>H NMR and <sup>19</sup>F NMR of compound 19.''' | |||
<html><A name="NMR19H"/></html>[[Image:Biomod_Aarhus_Chem_NMR_19H.png|900x900px|Center|thumb|<sup>1</sup>H NMR of compound 19]] | |||
<html><br style="clear:both;"/></html> | |||
<html><A name="NMR19F"/></html>[[Image:Biomod_Aarhus_Chem_NMR_19F.png|900x900px|Center|thumb|<sup>19</sup>F NMR of compound 19]] | |||
<html><br style="clear:both;"/></html> | |||
[[Image:Biomod_Aarhus_Chem_NMR_21H.png| | '''<sup>1</sup>H NMR and <sup>19</sup>F NMR of compound 21.''' | ||
[[Image:Biomod_Aarhus_Chem_NMR_21F.png| | |||
<html><A name="NMR21H"/></html>[[Image:Biomod_Aarhus_Chem_NMR_21H.png|900x900px|Center|thumb|<sup>1</sup>H NMR of compound 21]] | |||
<html><br style="clear:both;"/></html> | |||
<html><A name="NMR21F"/></html>[[Image:Biomod_Aarhus_Chem_NMR_21F.png|900x900px|Center|thumb|<sup>19</sup>F NMR of compound 21]] | |||
<html><br style="clear:both;"/></html> | |||
===Peptide lock=== | ===Peptide lock=== | ||
| Line 130: | Line 193: | ||
=====(S)-2-((tert-butoxycarbonyl)amino)-5-methoxy-5-oxopentanoic acid===== | =====(S)-2-((tert-butoxycarbonyl)amino)-5-methoxy-5-oxopentanoic acid===== | ||
<sup>1</sup>H NMR | <sup>1</sup>H NMR | ||
[[Image:Exp4HNMRfinishedforrealz | [[Image:Exp4HNMRfinishedforrealz.png|center|thumb|900x900px|<sup>1</sup>H NMR of (S)-2-((tert-butoxycarbonyl)amino)-5-methoxy-5-oxopentanoic acid]] | ||
<html><br style="clear:both;"/></html> | |||
<sup>13</sup>C NMR | <sup>13</sup>C NMR | ||
[[Image:Exp4CNMRfinishedforrealz | [[Image:Exp4CNMRfinishedforrealz.png|center|thumb|900x900px|<sup>13</sup>C NMR of (S)-2-((tert-butoxycarbonyl)amino)-5-methoxy-5-oxopentanoic acid]] | ||
<html><br style="clear:both;"/></html> | |||
For questions regarding raw data, please contact us by [[Biomod/2013/Aarhus/Supplementary/Team_And_Acknowledgments|email]]. | |||
{{Biomod/2013/Aarhus/Nano Creators/endhax}} | {{Biomod/2013/Aarhus/Nano Creators/endhax}} | ||
{{Biomod/2013/Aarhus/Nano_Creators/footer}} | {{Biomod/2013/Aarhus/Nano_Creators/footer}} | ||
Revision as of 07:43, 26 October 2013
<html> <style> /* ul.menu li.</html>Supplementary/Supplementary Data<html> a {
color: cyan;
}
- /
- toc {
display: none; }
- mytoc {
background: none; width: 200px; }
.toc { border: 0px solid; }
- toc ul ul,.toc ul ul {
margin: 0 0 0 1em; }
table.toc { background-color: #f0f4f4; }
- mytoc a,#mytoc a:visited {
font-size: normal; color: #222222; }
- mytoc a:hover {
font-color: #009ee0; /* text-decoration: underline; */ }
- wiki-toc {
width: 200px; margin-top: 6px; float: left; }
- wiki-body {
margin-left: 200px; padding-left: 12px; padding-right: 35px; }
- toc #toctitle,.toc #toctitle,#toc .toctitle,.toc .toctitle {
text-align: left; }
- toc h2,.toc h2 {
font-weight: normal; font-size: 17px; }
/*
- wiki-contents A {
color: #00aeef; }
- wiki-contents A:HOVER {
color: #00aeef; }
- /
- toctitle span {
display: none; }
/*
- wiki-body p,#wiki-body li,#wiki-body dd,div.thumbcaption {
font-size: medium; }
- /
/* required to avoid jumping */
- tocScrolWrapper {
/* left: 450px; */ position: absolute; /* margin-left: 35px; width: 280px; */ }
- tocScrol {
position: absolute; top: 0; /* just used to show how to include the margin in the effect */ /*margin-top: 20px; */ /* border-top: 1px solid purple; */ /*padding-top: 19px;*/ }
- tocScrol.fixed {
position: fixed; top: 0; }
- editPageTxt {
text-align: left; padding-left: 15px; }
- editPageTxt P {
clear: both; }
- toPageTop {
float: left; position: relative; top: 18px; left: 13px; color: #d13f31; } </style>
<script type="text/javascript"> $(document).ready(function() { var parentTable = $("#toc").parent(); $('#mytoc').append($("#toc").first());
$('#mytoc').find("#toc").attr("id", ""); parentTable.closest('table').remove(); });
$(document).ready( function() { var top = $('#tocScrol').offset().top - parseFloat($('#tocScrol').css('marginTop').replace( /auto/, 0)); var nav = $('#tocScrol'); var max = $('#indexing').offset().top - nav.height();
$(window).scroll(function(event) { // what the y position of the scroll is var y = $(this).scrollTop();
if (y > top) { // && signs are html decoded thus this construction if (y >= max) { nav.removeClass('fixed'); nav.css({ position : 'absolute', top : max - top }); } else { nav.addClass('fixed'); nav.removeAttr('style'); } } else { nav.removeClass('fixed'); nav.removeAttr('style'); } }); }); </script> <html> <html> <div id="wiki-contents"> <div id="tocScrolWrapper"> <div id="tocScrol"> <div id="wiki-toc"> <a id="toPageTop" href="#">▲</a> <table id="mytoc" class="toc" summary="Contents"> </table> <div id="editPageTxt"> <p> [<a href="http://openwetware.org/index.php?title=Biomod/2013/Aarhus/</html>Supplementary/Supplementary Data<html>&action=edit">edit this page</a>] </p> </div> </div> </div> </div> <div id="wiki-body"> </html>
Supplementary data
Origami design

<html><br style="clear:both;"/></html>
Staple strand sequences for the plate see supplementary information.

<html><br style="clear:both;"/></html>

<html><br style="clear:both;"/></html>
Figure and staple strand sequences for the dome see supplementary information.
sisiRNA
Yields of RP-HPLC purifications of the modified strands
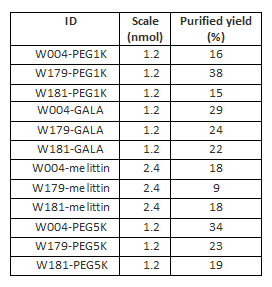
<html><br style="clear:both;"/></html>
Estimated yields of the annealing reactions

<html><br style="clear:both;"/></html>
Luciferase knockdown with all singly modified duplexes


<html><br style="clear:both;"/></html>

System in action
Radioactive labeling of W376

<html><br style="clear:both;"/></html> The band containing the labeled sample appears most similar to that in lane 4 which means that the 6 µL that were run on the gel contained 1.5 pmol W376, corresponding to a concentration of 0.25 µM in the purified sample.
Cholesterol
1H NMR, 13C NMR and IR of compound 4.
<html><A name="NMR4H"/></html>

<html><br style="clear:both;"/></html> <html><A name="NMR4C"/></html>

<html><br style="clear:both;"/></html>

<html><br style="clear:both;"/></html> <html><A name="IR4"/></html>

<html><br style="clear:both;"/></html> 1H NMR, 13C NMR and IR of compound 5.
<html><A name="NMR5H"/></html>

<html><br style="clear:both;"/></html> <html><A name="NMR5C"/></html>

<html><br style="clear:both;"/></html>

<html><br style="clear:both;"/></html> <html><A name="IR5"/></html>

<html><br style="clear:both;"/></html> 1H NMR and 13C NMR of compound 6.
<html><A name="NMR6H"/></html>

<html><br style="clear:both;"/></html> <html><A name="NMR6C"/></html>

<html><br style="clear:both;"/></html>

<html><br style="clear:both;"/></html>
Photosensitizer
1H NMR and 13C NMR of compound 7.
<html><A name="NMR7H"/></html>

<html><br style="clear:both;"/></html>
<html><A name="NMR7C"/></html>

<html><br style="clear:both;"/></html>
1H NMR and 13C NMR of compound 8.
<html><A name="NMR8H"/></html>

<html><br style="clear:both;"/></html>
<html><A name="NMR8C"/></html>

<html><br style="clear:both;"/></html>
1H NMR, 13C NMR and UV of compound 9.
<html><A name="NMR9H"/></html>

<html><br style="clear:both;"/></html>
<html><A name="NMR9C"/></html>

<html><br style="clear:both;"/></html>
<html><A name="9UV"/></html>  <html><br style="clear:both;"/></html>
<html><br style="clear:both;"/></html>
1H NMR, 13C NMR and UV of compound 10.
<html><A name="NMR10H"/></html>

<html><br style="clear:both;"/></html>
<html><A name="NMR10C"/></html>

<html><br style="clear:both;"/></html>
<html><A name="10UV"/></html>  <html><br style="clear:both;"/></html>
<html><br style="clear:both;"/></html>
1H NMR and 13C NMR of compound 11.
<html><A name="NMR11H"/></html>

<html><br style="clear:both;"/></html>
<html><A name="NMR11C"/></html>

<html><br style="clear:both;"/></html>
1H NMR and 13C NMR of compound 12.
<html><A name="NMR12H"/></html>

<html><br style="clear:both;"/></html>
<html><A name="NMR12C"/></html>

<html><br style="clear:both;"/></html>
5-propargylamino-ddUTP
1H NMR, 13C NMR and IR of compound 15.
<html><A name="NMR15H"/></html>

<html><br style="clear:both;"/></html>

<html><br style="clear:both;"/></html>
<html><A name="NMR15C"/></html>

<html><br style="clear:both;"/></html>
<html><A name="IR15"/></html>

<html><br style="clear:both;"/></html>
1H NMR, 13C NMR and IR of compound 16.
<html><A name="NMR16H"/></html>

<html><br style="clear:both;"/></html>

<html><br style="clear:both;"/></html>
<html><A name="NMR16C"/></html>

<html><br style="clear:both;"/></html>
<html><A name="IR16"/></html>

<html><br style="clear:both;"/></html>
1H NMR, 13C NMR and IR of compound 17.
<html><A name="NMR17H"/></html>

<html><br style="clear:both;"/></html>

<html><br style="clear:both;"/></html>
<html><A name="NMR17C"/></html>

<html><br style="clear:both;"/></html>
<html><A name="IR17"/></html>

<html><br style="clear:both;"/></html>
1H NMR, 13C NMR and IR of compound 18.
<html><A name="NMR18H"/></html>

<html><br style="clear:both;"/></html>

<html><br style="clear:both;"/></html>
<html><A name="NMR18C"/></html>

<html><br style="clear:both;"/></html>
<html><A name="IR18"/></html>

<html><br style="clear:both;"/></html>
1H NMR and 19F NMR of compound 19.
<html><A name="NMR19H"/></html>

<html><br style="clear:both;"/></html>
<html><A name="NMR19F"/></html>

<html><br style="clear:both;"/></html>
1H NMR and 19F NMR of compound 21.
<html><A name="NMR21H"/></html>

<html><br style="clear:both;"/></html>
<html><A name="NMR21F"/></html>

<html><br style="clear:both;"/></html>
Peptide lock
Spectra
(S)-2-((tert-butoxycarbonyl)amino)-5-methoxy-5-oxopentanoic acid
1H NMR

<html><br style="clear:both;"/></html>
13C NMR

<html><br style="clear:both;"/></html>
For questions regarding raw data, please contact us by email.
<html></div></div></html> <html> <head> <style>
- indexing {
/* float: left; position: center; */ background-color: #222; border-top: 2px solid #d13f31; color: #006e9c; margin: 0px; padding: 0px 0px 10px 0px; width: 100%; text-align: center; }
.footer-section { padding: 10px; display: table-cell; text-align: left; }
.footer-section-title { font-size: 20px; }
- footer-contents {
color: #006e9c; display: inline-table; }
.footer-section A { color: #006e9c; text-decoration: none; }
.footer-section A:HOVER { color: #00aeef; }
.footer-section ul { list-style-type: square; }
- sitemapTitle {
margin-top: 20px; font-size: 24px; }
- editFooter {
float: right; margin-top: -28px; margin-right: 5px; }
- editFooter A {
color: #006e9c; text-decoration: none; }
.cf:before,.cf:after { content: " "; /* 1 */ display: table; /* 2 */ }
.cf:after { clear: both; }
- bodyContent a[href^="mailto:"], .link-mailto {
background: url() no-repeat scroll right center transparent; padding-right: 0px; color: #006e9c;
}
</style> </head> <body> <div id="indexing"> <div id="sitemap"> <p id="sitemapTitle">SITEMAP | BIOMOD 2013 NANO CREATORS | Aarhus University</p> <div id="footer-contents"> <div class="footer-section"> <p class="footer-section-title">INTRODUCTION</p> <ul> <li><a href="/wiki/Biomod/2013/Aarhus">Home, abstract, animation and video</a></li> <li><a href="/wiki/Biomod/2013/Aarhus/Introduction">Introduction</a></li </ul> </div> <div class="footer-section"> <p class="footer-section-title">RESULTS AND DISCUSSION</p> <ul> <li><a href="/wiki/Biomod/2013/Aarhus/Results_And_Discussion/Origami">Origami</a></li> <li><a href="/wiki/Biomod/2013/Aarhus/Results_And_Discussion/Peptide_lock">Peptide lock</a></li> <li><a href="/wiki/Biomod/2013/Aarhus/Results_And_Discussion/Chemical_Modification">Chemical modification</a></li> <li><a href="/wiki/Biomod/2013/Aarhus/Results_And_Discussion/sisiRNA">sisiRNA</a></li> <li><a href="/wiki/Biomod/2013/Aarhus/Results_And_Discussion/System_In_Action">System in action</a></li> </ul> </div> <div class="footer-section"> <p class="footer-section-title">MATERIALS AND METHODS</p> <ul> <li><a href="/wiki/Biomod/2013/Aarhus/Materials_And_Methods/Origami">Origami</a></li> <li><a href="/wiki/Biomod/2013/Aarhus/Materials_And_Methods/Peptide_lock">Peptide lock</a></li> <li><a href="/wiki/Biomod/2013/Aarhus/Materials_And_Methods/Chemical_Modification">Chemical modification</a></li> <li><a href="/wiki/Biomod/2013/Aarhus/Materials_And_Methods/sisiRNA">sisiRNA</a></li> <li><a href="/wiki/Biomod/2013/Aarhus/Materials_And_Methods/System_In_Action">System in action</a></li> <li><a href="/wiki/Biomod/2013/Aarhus/Materials_And_Methods/Methods">Methods</a></li> </ul> </div> <div class="footer-section"> <p class="footer-section-title">SUPPLEMENTARY</p> <ul> <li><a href="/wiki/Biomod/2013/Aarhus/Supplementary/Team_And_Acknowledgments">Team and acknowledgments</a></li> <li><a href="/wiki/Biomod/2013/Aarhus/Supplementary/Optimizations">Optimizations</a></li> <li><a href="/wiki/Biomod/2013/Aarhus/Supplementary/Supplementary_Data">Supplementary data</a></li>
<li><a
href="/wiki/Biomod/2013/Aarhus/Supplementary/Supplementary_Informations">Supplementary informations</a> <li><a href="/wiki/Biomod/2013/Aarhus/Supplementary/References">References</a></li> </ul> </div> </div> <div> <p id="copyright">Copyright (C) 2013 | BIOMOD Team Nano Creators @ Aarhus University | Programming by: <a href="mailto:pvskaarup@gmail.com?Subject=BIOMOD 2013:">Peter Vium Skaarup</a>.</p> </div> </div>
<!-- Sponsers --> <div> <img alt="Sigma - Aldrich" src="http://openwetware.org/images/3/39/Sigmaaldrich-logo%28transparant%29.png" width="300px" height="154px"> <img alt="VWR International" src="http://openwetware.org/images/2/28/Vwr_logo.png" width="300px" height="61px"> <img alt="Promega" src="http://openwetware.org/images/7/72/Promega.png" width="175px" height="105px" style="padding-right: 5px; padding-left: 5px;"> <img alt="kem-en-tec" src="http://openwetware.org/images/3/3a/Kementec.png" width="130px" height="129px"> <img alt="Centre For Dna Nanotechnology" src="http://openwetware.org/images/4/4f/CDNA_logo.png" width="420px" height="90px"> <img alt="Dansk Tennis Fond" src="http://openwetware.org/images/9/9a/Dansk_tennis.png" width="250px" height="53px"> </div> <div id="editFooter"> [<a href="http://openwetware.org/index.php?title=Template:Biomod/2013/Aarhus/Nano_Creators/footer&action=edit">edit sitemap</a>] </div> </div> </body> </html>