BME494 Sp2014 Dhatt: Difference between revisions
No edit summary |
|||
| (36 intermediate revisions by the same user not shown) | |||
| Line 27: | Line 27: | ||
<!-- Below is a template code for making a thumbnail image to embed in your text. See BME494_Wiki_Help for help with uploading images to OpenWetWare and using them on your page.--> | <!-- Below is a template code for making a thumbnail image to embed in your text. See BME494_Wiki_Help for help with uploading images to OpenWetWare and using them on your page.--> | ||
[[Image:BME494_Dhatt_ClassicalDesignGardner.jpg|thumb| | [[Image:BME494_Dhatt_ClassicalDesignGardner.jpg|thumb|440px||left|Classical Toggle Switch Design (Gardner et al, 2000)]] | ||
| Line 35: | Line 35: | ||
<!-- Incorporate information from Presentation 1. Must be reasonable and clearly relevant to an important health challenge. --> | <!-- Incorporate information from Presentation 1. Must be reasonable and clearly relevant to an important health challenge. --> | ||
Diabetes mellitus, or diabetes, is a group of metabolic diseases in which an individual has high blood sugar. Diabetes occurs due to the either the pancreas not producing enough insulin or because cells of the body do not respond to the insulin being produced. There are three types of diabetes: Type I, Type II, and gestational diabetes. Serious long-term complications of diabetes include heart disease, kidney failure, and damage to the eyes. Diabetes is a chronic disease and there is no known preventative measure for type I diabetes. Having awareness of the disease and treating it at early stages can prevent and lower the risk of complications in patients with diabetes. Patients with diabetes must maintain keeping both short-term and long-term blood glucose levels within acceptable ranges. | |||
Globally, 227-285 million individuals have diabetes and about 90% of these individuals have Type II diabetes. In 2011, 1.4 million deaths occurred worldwide due to the result of diabetes making it the 8th leading cause of death. This number is estimated to double in the next 15 years, therefore, there is a need for a rapid diagnostic test. | |||
<br><br> | |||
<!-- These are lines breaks for spacing purposes. You can add or delete these as needed --> | <!-- These are lines breaks for spacing purposes. You can add or delete these as needed --> | ||
==Design of a New Device== | ==Design of a New Device== | ||
The device will be designed as a diagnostic tool for diabetes and the functionality of the genetic switch replicates that of an “AND” logic gate. The device will require two conditions to be true in order for an output to be produced. One condition is that IPTG must be present in the device’s environment. When IPTG is present, it will bind to the LacI repressor and allow for transcription to continue. The other condition is that glucose levels in the device’s environment must be low. Glucose levels affect production of cAMP inversely; when glucose levels are high, cAMP production decreases and when glucose levels are low, cAMP production increases. cAMP binds to catabolite activator protein (CAP_ to form the CAP-cAMP complex. For the complex, cAMP must be present and glucose levels must be low. This complex is the required input of the device. In the natural lac operon, the CAP-cAMP complex leads to activation of gene expression from the lac operon. If glucose is present, cAMP levels will be low and the host will metabolize glucose if lactose is present. | |||
<!-- Show a network/ circuit diagram of your device. Include a paragraph to explain how it works (e.g., how to switch the system from on to off and vice versa, and what happens to each component as the system switches between states) --> | <!-- Show a network/ circuit diagram of your device. Include a paragraph to explain how it works (e.g., how to switch the system from on to off and vice versa, and what happens to each component as the system switches between states) --> | ||
[[Image: | [[Image:BME494_Dhatt_AndGate.jpg|500px|AND gate logic gene toggle switch. IPTG and low glucose levels conditions must be met in order for GFP production]] | ||
<br>AND gate logic gene toggle switch. IPTG and low glucose levels conditions must be met in order for GFP production.<br> | |||
[[Image:BME494_Dhatt_AndGateTable.jpg|500px|Table describing that both inputs are needed in order to produce an output]] | |||
<br>Table describing that both inputs are needed in order to produce an output.<br> | |||
<br><br> | |||
<!-- These are lines breaks for spacing purposes. You can add or delete these as needed --> | <!-- These are lines breaks for spacing purposes. You can add or delete these as needed --> | ||
| Line 59: | Line 64: | ||
'''SYNTHETIC DNA LAYOUT''' | '''SYNTHETIC DNA LAYOUT''' | ||
Type IIs Assembly was used to build this lac switch. Type IIs Assembly allows for parts to be assembled in one step. For Type IIs Assembly, forward and reverse primers are needed to be created and placed in the system in order to create sticky ends that can bind various parts together. To put the pieces together, PCR is implemented, which allows all the parts to be replicated thousands of times in order to produce a desired final product. Digestion and ligation is used during which BsmBI cuts the DNA fragments and creates complementary overhangs that anneal via base pairing. | |||
[[Image:BME494_Dhatt_Assembly.jpg|500px|center]] | |||
<!-- Show each DNA fragment in the order in which you propose to assemble them into a single plasmid. This can be a series of connected blocks. You do not have to show the plasmid as a circle. --> | <!-- Show each DNA fragment in the order in which you propose to assemble them into a single plasmid. This can be a series of connected blocks. You do not have to show the plasmid as a circle. --> | ||
'''RESOURCES''' | '''RESOURCES''' | ||
<!-- List pre-existing parts/ Biobricks that you will use. Also list new parts that you will need to get from a natural source. BE SPECIFIC. --> | <!-- List pre-existing parts/ Biobricks that you will use. Also list new parts that you will need to get from a natural source. BE SPECIFIC. --> | ||
The following BioBrick parts, found on the iGEM registry website, will be used to build the new device:<br> | |||
K418003 - composite Lac promoter inducible by IPTG<br> | |||
Size: 1416 base pairs<br> | |||
LacI present: transcription inhibited<br> | |||
LacI absent: transcription promoted<br> | |||
LacI inhibited by IPTG<br> | |||
B0030 – Strong RBS<br> | |||
Size: 15 base pairs<br> | |||
K259006 – Composite part made from green fluorescent protein (GFP) and double terminator<br> | |||
IPTG present: GFP produced<br> | |||
IPTG absent: GFP not produced, clear solution<br> | |||
Size: 823 base pairs<br> | |||
pSB1A3 – BioBrick assembly plasmid<br> | |||
Size:2155 base pairs<br> | |||
'''TYPE IIS ASSEMBLY''' | '''TYPE IIS ASSEMBLY''' | ||
'''PCR'''<br> | |||
Polymerase Chain Reaction (PCR) will be used for amplification of the DNA parts. PCR is the process of adding DNA, primers, nucleotides and DNA polymerase to a tube which produces system through assembly pot process after placed in a thermocycler and placed through many cycles. PCR consists of three specific temperature steps: denaturation, annealing, and elongation. Denaturation allows the DNA template to be heated to a specific temperature and yield single-stranded DNA molecules. The reaction temperature is then lowered for a short period of time in order to allow the complementary DNA primers to anneal to the single-stranded DNA template. The primers are then elongated by the DNA polymerase that is used. The DNA polymerase then allows for the cycle to be repeated multiple times resulting in thousands of copies of the desired fragments. | |||
'''Digestion/Ligation Reaction'''<br> | |||
The digestion/ligation reaction allows for dilution the purified PCR products to volume of 20 ul. The digestion/ligation reaction uses BsmB1, a Type II restriction enzyme, which cuts DNA fragments to create complementary overhangs (sticky ends). These sticky ends anneal via DNA base pairs. Primers introduce these complementary overhangs to the DNA. T4 ligase is used to seal the gaps between base pairs and create a finished system. | |||
'''Primers'''<br> | |||
The following primers will be used for the new device in order to introduce complementary overhangs and BsmBI cut sites.<br> | |||
Forward Primer Vector: 5'-cacaccaCGTCTCaactagtagcggccgct<br> | |||
Reverse Primer Vector: 5'-cacaccaCGTCTCatctagatgcggccgcg<br> | |||
Forward Primer Composite Lac Promoter: 5'-cacaccaCGTCTCatagattgacagctagctca<br> | |||
Reverse Primer Composite Lac Promoter: 5'-cacaccaCGTCTCatgtgtgtgctcagtatctt<br> | |||
Forward Primer RBS: 5'-cacaccaCGTCTCattaaagaggagaaa<br> | |||
Reverse Primer RBS: 5'-cacaccaCGTCTCaaattttctcctctttaat<br> | |||
Forward Primer GFP with terminator: 5'-cacaccaCGTCTCaatgcgtaaaggagaa<br> | |||
Reverse Primer GFP with terminator: 5'-cacaccaCGTCTCatagtaaataataaaaaagc<br> | |||
Forward Primer for the mutation: 5’-agctgttgccGgtctcactgg<br> | |||
Reverse Primer for the mutation: 5’-ccagtgagacCggcaacagct<br> | |||
'''Reagents'''<br> | |||
PCR Reagents:<br> | |||
{| border="1" cellpadding="5" style="width: 100%" class="wikitable" | |||
|+ '''PCR Reagents''' | |||
! Reagent | |||
! Volume | |||
|- | |||
! Template DNA | |||
| 0.2 μL | |||
|- | |||
! 1.0 uM Forward Primer | |||
| 1.0 μL | |||
|- | |||
! 1.0 uM Reverse Primer | |||
| 1.0 μL | |||
|- | |||
! 2x GoTaq Green Mix | |||
| 25 μL | |||
|- | |||
! H<sub>2</sub>O | |||
| 22.8 μL | |||
|- | |||
! TOTAL VOLUME | |||
| 50 μL | |||
|- | |||
|}<br> | |||
The PCR reagents used the following thermal cycling properties: <br> | |||
95°C, 3 min.<br> | |||
[95°C, 15 sec; 55°C, 15 sec; 72°C, 30 sec] x30<br> | |||
72°C, 3 min.<br> | |||
4°C, ∞<br> | |||
<!-- Incorporate information from Presentation 2 --> | <!-- Incorporate information from Presentation 2 --> | ||
Digestion/Ligation Reaction Reagents:<br> | |||
{| border="1" cellpadding="5" style="width: 100%" class="wikitable" | |||
|+ '''Digestion/Ligation Reagents''' | |||
! Reagent | |||
! Assembly | |||
! Negative Control | |||
|- | |||
| PCR fragment 1 (vector) | |||
| 1.0 μL | |||
| 1.0 μL | |||
|- | |||
| PCR fragment 2 | |||
| 1.0 μL | |||
| None | |||
|- | |||
| PCR fragment 3 | |||
| 1.0 μL | |||
| None | |||
|- | |||
| PCR fragment 4 | |||
| 1.0 μL | |||
| None | |||
|- | |||
| PCR fragment 5 | |||
| 1.0 μL | |||
| None | |||
|- | |||
| 10x T4 Ligase | |||
| 0.25 μL | |||
| 0.25 μL | |||
|- | |||
| BsmB1 | |||
| 0.5 μL | |||
| 0.5 μL | |||
|- | |||
| H<sub>2</sub>O | |||
| 4.25 μL | |||
| 8.25 μL | |||
|- | |||
| Total Volume | |||
| 10 μL | |||
| 10 μL | |||
|- | |||
|}<br> | |||
The Digestion/Ligation reagents used the following thermal cycling properties: <br> | |||
[45°C, 2 min.; 16°C, 5 min] x25<br> | |||
60°C, 10 min. | |||
80°C, 20 min.<br> | |||
4°C, ∞<br> | |||
<!-- Incorporate information from Presentation 2 --> | |||
<br><br><br> | |||
<!-- These are lines breaks for spacing purposes. You can add or delete these as needed --> | <!-- These are lines breaks for spacing purposes. You can add or delete these as needed --> | ||
| Line 79: | Line 223: | ||
'''LAC OPERON MODEL SIMULATION''' | '''LAC OPERON MODEL SIMULATION''' | ||
<br> | <br> | ||
I used a model of the natural Lac operon to learn how changing the parameter values changes the behavior of the system. | I used a model of the natural Lac operon to learn how changing the parameter values changes the behavior of the system.<br> | ||
<!-- Continue this paragraph by explaining how you interacted with the MatLab model. Include two or more images showing different output curves that were generated when you altered the IPTG input concentration --> | <!-- Continue this paragraph by explaining how you interacted with the MatLab model. Include two or more images showing different output curves that were generated when you altered the IPTG input concentration --> | ||
<br> | |||
[[Image:BME494_Dhatt_MRNA0.25.jpg|400px|]] | |||
[[Image:BME494_Dhatt_MRNA0.35.jpg|400px|]] | |||
<br> | <br> | ||
[[Image:BME494_Dhatt_Bgal0.25.jpg|400px|]] | |||
[[Image:BME494_Dhatt_Bgal0.35.jpg|400px|]] | |||
'''RELATIONSHIP BETWEEN THE LAC MODEL AND MY NEW DESIGN''' | |||
<br> | <br> | ||
At an IPTG concentration of 0.25M, the figure shows that mRNA concentration decreased and at a concentration of 0.35M, the figure below shows that concentration of mRNA increased. Concentraion of Bgal increased over time as concentration of IPTG was increased. <br> | |||
<br> | |||
'''Similarities''': <!-- Describe what aspects of the Lac operon model are related to your system --> | |||
The Lac operon is useful to the device since the device also uses LacI and can be useful to see how the different concentrations of IPTG affect the system. | |||
<br> | <br> | ||
<!-- | '''Differences''': <!-- Describe what aspects of your new device are not accounted for by the Lac operon model, and how this difference would require changes or additions to the Lac operon model. You may write this in general terms if you are not yet familiar with advanced biological modeling. Try your best! ---> | ||
The Lac operon model does not take into account glucose levels, which is an important component of the device and the and-logic gate. A model that incorporates both Lactose and glucose levels would be essential in order to test the device. | |||
<br><br> | |||
==Human Practices== | |||
[[Image:cow.jpg|thumb|130px|right|'''Danger of Chemicals in Farmlands''']] | |||
<!--Wait until Unit 3 to fill this in. Demonstrate that you have considered the societal aspects of your project. --> | |||
<br> | '''SUPPORTS AND UNDERSTANDS'''<br> | ||
The demographic consists of individuals who understand the scientific nature of the device. Individuals who understand the risk involved using E.Coli and creating a gene system. This group would most likely consist of researchers, scientists, physicians, and biologists. | |||
'''DOES NOT SUPPORT AND UNDERSTANDS'''<br> | |||
This demographic group understands the background of the device and the scientific nature but do not support it. This group may consist of individuals who are aware of devices that act as diagnostic tests for diabetes and also individuals who are aware of the scientific nature of the device but do not believe in the application. | |||
'''SUPPORTS BUT DOES NOT UNDERSTAND'''<br> | |||
This group consists of individuals who support the research of the new device used to diagnose diabetes but may not understand the scientific nature of the device and its scientific application. These individuals may include those who understand the need of the device from a market perspective where there is a need for a low-cost, rapid device. | |||
< | '''DOES NOT SUPPORT NOR UNDERSTANDS'''<br> | ||
This demographic group nor understands the scientific background of the device or supports its application. These individuals would include activists who do not support synthetic biology and the application or genetic systems which, therefore, forces them to not understand the scientific benefit of it to society. | |||
<br><br> | |||
==About the Designer== | ==About the Designer== | ||
| Line 119: | Line 278: | ||
<div style="color: #808080; background-color: #ffffff; width: 600px; padding: 5px"> | <div style="color: #808080; background-color: #ffffff; width: 600px; padding: 5px"> | ||
[[Image: | [[Image:BME494_Dhatt_profile.jpg |thumb|noframe|130px|left|'''Kulveen Dhatt''']] | ||
* My name is | * My name is Kulveen, and I am a senior majoring in Biomedical Engineering . I am taking BME 494 because synthetic biology sounded extremely interesting and it was a new topic of interest to me. An interesting fact about me is that I was born in India and moved here with my parents at a very young age.. | ||
</div> | </div> | ||
<br> | |||
==References== | ==References== | ||
<br> | |||
1. "American Diabetes Association". http://www.diabetes.org/<br> | |||
2. "Haynes:TypeIIS Assembly". http://openwetware.org/wiki/Haynes:TypeIIS_Assembly<br> | |||
3. Gardner T.S., Cantor C.Reverse, Collins J.J. 2000. Nature. Vol 409: 339-342.<br> | |||
4. iGEM Registry of Standard Biological Parts. http://parts.igem.org/Main_Page<br> | |||
Latest revision as of 17:13, 8 May 2014
My Profile Dr. Haynes OpenWetWare Previous Course Wiki Editing Help
Background & Proposed ApplicationBACKGROUND
The synthetic system modeled in the “Construction of a genetic toggle switch In Escherichia coli” uses two different states, an “on” state” and an “off” state (Gardner et all 2000). The classical system uses two repressor and two promoter pairs in which both repressors are inhibited by a different inducer, aTc and IPTG. The promoters were placed next to the repressor gene of the opposite pair. This system represents a bistable gene-regulatory network where only one promoter can be expressed at one time because the expression of one promoter repressed expression of the other promoter. The on-state of the cell was represented by placing a green fluorescent protein (GFP) transcription gene downstream of the on-state promoter. 
Globally, 227-285 million individuals have diabetes and about 90% of these individuals have Type II diabetes. In 2011, 1.4 million deaths occurred worldwide due to the result of diabetes making it the 8th leading cause of death. This number is estimated to double in the next 15 years, therefore, there is a need for a rapid diagnostic test.
Design of a New DeviceThe device will be designed as a diagnostic tool for diabetes and the functionality of the genetic switch replicates that of an “AND” logic gate. The device will require two conditions to be true in order for an output to be produced. One condition is that IPTG must be present in the device’s environment. When IPTG is present, it will bind to the LacI repressor and allow for transcription to continue. The other condition is that glucose levels in the device’s environment must be low. Glucose levels affect production of cAMP inversely; when glucose levels are high, cAMP production decreases and when glucose levels are low, cAMP production increases. cAMP binds to catabolite activator protein (CAP_ to form the CAP-cAMP complex. For the complex, cAMP must be present and glucose levels must be low. This complex is the required input of the device. In the natural lac operon, the CAP-cAMP complex leads to activation of gene expression from the lac operon. If glucose is present, cAMP levels will be low and the host will metabolize glucose if lactose is present.
Building the New DeviceSYNTHETIC DNA LAYOUT Type IIs Assembly was used to build this lac switch. Type IIs Assembly allows for parts to be assembled in one step. For Type IIs Assembly, forward and reverse primers are needed to be created and placed in the system in order to create sticky ends that can bind various parts together. To put the pieces together, PCR is implemented, which allows all the parts to be replicated thousands of times in order to produce a desired final product. Digestion and ligation is used during which BsmBI cuts the DNA fragments and creates complementary overhangs that anneal via base pairing. 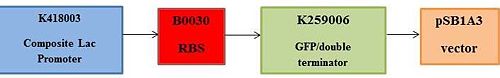
The following BioBrick parts, found on the iGEM registry website, will be used to build the new device: K418003 - composite Lac promoter inducible by IPTG
PCR Polymerase Chain Reaction (PCR) will be used for amplification of the DNA parts. PCR is the process of adding DNA, primers, nucleotides and DNA polymerase to a tube which produces system through assembly pot process after placed in a thermocycler and placed through many cycles. PCR consists of three specific temperature steps: denaturation, annealing, and elongation. Denaturation allows the DNA template to be heated to a specific temperature and yield single-stranded DNA molecules. The reaction temperature is then lowered for a short period of time in order to allow the complementary DNA primers to anneal to the single-stranded DNA template. The primers are then elongated by the DNA polymerase that is used. The DNA polymerase then allows for the cycle to be repeated multiple times resulting in thousands of copies of the desired fragments.
Forward Primer Vector: 5'-cacaccaCGTCTCaactagtagcggccgct Forward Primer Composite Lac Promoter: 5'-cacaccaCGTCTCatagattgacagctagctca Forward Primer RBS: 5'-cacaccaCGTCTCattaaagaggagaaa Forward Primer GFP with terminator: 5'-cacaccaCGTCTCaatgcgtaaaggagaa Forward Primer for the mutation: 5’-agctgttgccGgtctcactgg
Reagents
The PCR reagents used the following thermal cycling properties:
The Digestion/Ligation reagents used the following thermal cycling properties:
Testing the New DeviceLAC OPERON MODEL SIMULATION
RELATIONSHIP BETWEEN THE LAC MODEL AND MY NEW DESIGN
Differences: The Lac operon model does not take into account glucose levels, which is an important component of the device and the and-logic gate. A model that incorporates both Lactose and glucose levels would be essential in order to test the device.
Human Practices
DOES NOT SUPPORT AND UNDERSTANDS SUPPORTS BUT DOES NOT UNDERSTAND DOES NOT SUPPORT NOR UNDERSTANDS
About the Designer
References
|






