BME103:T930 Group 3: Difference between revisions
Rohan Kumar (talk | contribs) |
Rohan Kumar (talk | contribs) |
||
| Line 94: | Line 94: | ||
[[Image:BME-03 box.jpg|200px|The exterior of the fluorimeter]] | [[Image:BME-03 box.jpg|200px|The exterior of the fluorimeter]] | ||
[[Image:BME003 Part1.jpg|200px|The interior. Samsung camera facing the slide]] | [[Image:BME003 Part1.jpg|200px|The interior. Samsung camera facing the slide]] | ||
[[Image:BME003Part3.jpg|200px|The interior. close up camera]] | [[Image:BME003Part3.jpg|200px|The interior. close up camera]]<br> | ||
1.) The first picture is the extertor of the fuorimeter | 1.) The first picture is the extertor of the fuorimeter | ||
2.) The second picture shows the interior. The smartphone camera is angled to have a view of the slide. During actual use the slide platform was sightly elevated with plates for a better view of the drop. | 2.) The second picture shows the interior. The smartphone camera is angled to have a view of the slide. During actual use the slide platform was sightly elevated with plates for a better view of the drop. | ||
Revision as of 23:54, 14 November 2012
| Home People Lab Write-Up 1 Lab Write-Up 2 Lab Write-Up 3 Course Logistics For Instructors Photos Wiki Editing Help | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
OUR TEAM
LAB 1 WRITE-UPInitial Machine TestingThe Original Design
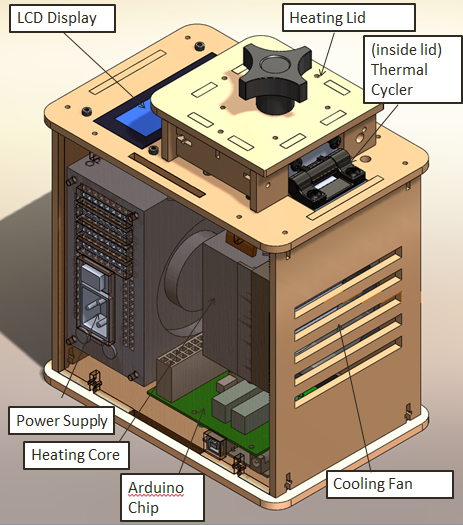
When we unplugged the lcd display wire(part 3) from the Arudnio chip(part 6), the screen turned off. Everything on the PCR was working fine expect there was no output on the display. When we unplugged the white wire that connects Arudino chip(part 6) to thermal cycler (part 2), the reading from the screen dropped to -40 degrees Celsius. We disconnected the wire multiple times and each time the screen displayed -40 degrees Celsius.
We ran a test run on 10/25/2012. For this test we placed some empty PCR tubes into the machine and ran a simple test program on the Open PCR software. After the simple test was over we noticed that the display screen on the Open PCR lid matched very closely with what was displayed on our computer screen. The agreement between our computer screen and our PCR display meant that our diagnostic test was a success.
ProtocolsPolymerase Chain Reaction Polymerase Chain Reaction Procedure: 2.)We labeled 8 empty PCR tubes. For the first sample we labeled the 3 DNA samples 1A, 1B and 1C. For the second sample we labeled the tubes 2A, 2B and 2C. For the positive and negative controls, we labeled the tubes + and - respectively. 3.)Using one pipette per sample, to avoid contamination, we transferred the PCR reaction mix we were given to the PCR tubes. 4.)We then placed the samples in the PCR machine 5.)We set our PCR program to three stages. Stage one: 1 cycle, 95 degree Celsius for 3 minutes. Stage 2: 35 cycles, 95 degrees Celsius for 30 seconds, 57 degrees Celsius for 30 seconds, 72 degrees Celsius. Stage three: 72 degrees Celsius for 3 minutes and then hold at 4 degree Celsius.
Sample one ID 43891: 48 Male GO Taq DNA Mix
Fluorimeter Assembly
Fluorimeter Procedure: 2.)With the permanent marker we also labeled the Eppendrof tubes at the top, we had a total of 10 Eppendrof tubes labeled and 10 pipettes labeled. 3.)We transferred each sample separately into the Eppendorf tubes containing 400 ml of buffer. 4.)Using a specially labeled Eppendorf tube containing SYBR GREEN, with its own pippter, we placed two drops onto the first two center drops. 5.)Then using the sample we placed two drops on top of the SYBR GREEN solution drops 6.)Then we aligned the blue light to pass through the drop. 7.)Then the smartphone operator took a picture with the settings on the phone adjusted to inactive flash, iso to 800, white balance to auto, exposure to the highest setting and contrast to the lowest setting. 8.)This process was repeated for all samples 9.)After picture was taken it was given to the Image J software Image J Procedure: 2.)Using the menu selection we used, image > color > split channels 3.) This created 3 files 4.)The we clicked menu bar to activate the oval selection. 5.)We drew an oval around our green drop image and then selected analyze > measure. 6.) We then repeated the oval process but for the area above the drop, to get the noise measurement.
Research and DevelopmentSpecific Cancer Marker Detection - The Underlying Technology (Add a write-up of the information discussed in Week 3's class) (BONUS points: Use a program like Powerpoint, Word, Illustrator, Microsoft Paint, etc. to illustrate how primers bind to the cancer DNA template, and how Taq polymerases amplify the DNA. Screen-captures from the OpenPCR tutorial might be useful. Be sure to credit the source if you borrow images.)
Results
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||